In global beauty manufacturing, “scale” is never a single order. It is a controlled progression — from early trial batches to repeat orders, from one product format to a multi-SKU portfolio, and from basic execution to a repeatable packaging system capable of surviving fast launches, channel expansion, and relentless cost pressure.
This is the story of how a family-operated Eastern European beauty business did exactly that. Starting with modest 10,000-unit batches of roll-on bottles, the brand evolved into a high-volume perfume program exceeding one million units annualized — backed by formal quality gates, disciplined pre-production sampling, and a supplier relationship built on genuine accountability.
This case study documents each phase of that journey in full operational detail. Whether you are a brand owner, OEM filling factory, or regional beauty trader scaling your own packaging system, the principles inside this story are directly applicable to your next program.
Who Is the Client?
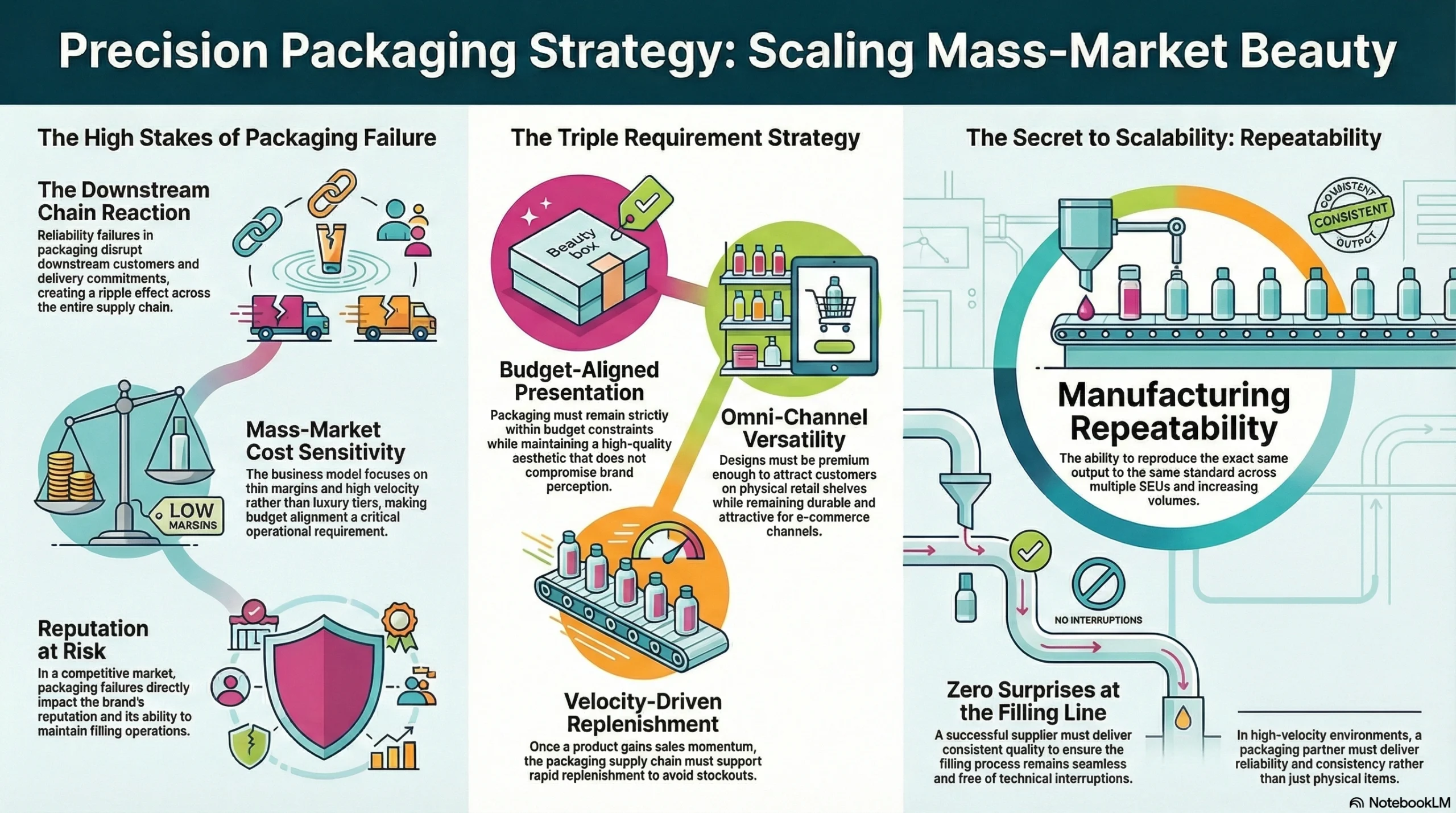
The client is a family-operated beauty business that combines skincare and fragrance packaging into one portfolio. Their business model includes OEM and ODM manufacturing alongside filling operations — which means every packaging reliability failure on their end doesn’t just affect them. It creates a chain reaction that disrupts their downstream customers, their delivery commitments, and ultimately their reputation in a competitive market.
Their positioning was consistently mass-market and cost-sensitive. They were not chasing luxury tiers. Their requirement was sharply defined from the very beginning:
Packaging must remain budget-aligned without compromising presentation
Packaging must look premium enough for both shelf and e-commerce channels
Packaging must support fast replenishment once a product gains proven sales momentum
This is exactly the environment that exposes weak suppliers. When margins are thin and velocity matters, a packaging partner must deliver far more than products. They must deliver repeatability — the ability to reproduce the same output, to the same standard, at increasing volumes, across multiple SKUs, with zero surprises at the filling line.
How the Relationship Grew: Three Shifts That Defined the Scale-Up
At the start, the client purchased small batches — commonly around 10,000 units per SKU — of relatively standard packaging formats. What changed over time was not just volume. Three structural shifts occurred that redefined the entire relationship:
From single items → to coordinated SKUs: multiple designs, multiple components, managed together
From “looks good” → to “passes tests”: adhesion testing, vacuum testing, fitment controls, and cosmetic defect management became program standards
From one category → to multi-category scaling: roll-on bottles, dropper bottles, cream jars, and full perfume systems, all running in parallel
The milestone outcome: the partnership progressed into a perfume bottle program that grew from a large first production run into an annualized path exceeding one million units — with forward planning discussions already underway for doubling volume again.
Phase 1: Establishing Repeatability with Small Batches
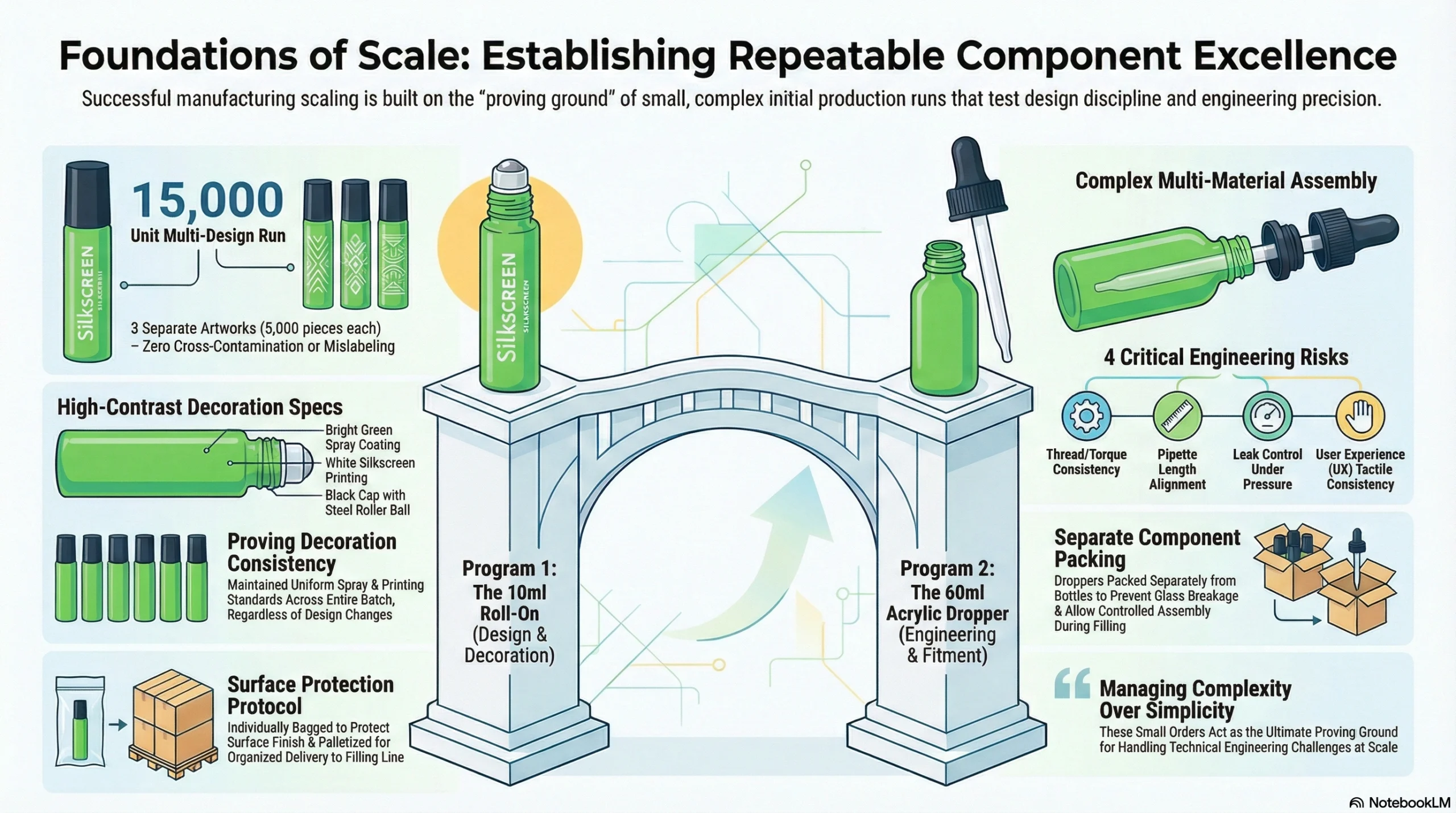
10ml Roll-On Bottle Program — Decorated, Multi-Design
The earliest stage of the relationship focused on small, standard orders. But “small” did not mean simple. These orders carried operational rules that would become the foundation for everything that followed.
The client placed an order for a 10ml roll-on bottle combining decoration and design variation across the same production run.
Key specifications:
| Parameter | Detail |
|---|---|
| Coating | Bright green spray coating |
| Printing | White silkscreen |
| Design Allocation | 3 designs × 5,000 pcs each (total 15,000 pcs) |
| Components | Black cap + steel roller ball + semi-transparent ball holder |
| Pre-Production Samples | 5 sets required |
| Packing | Individually bagged; palletized shipment |
Design allocation discipline — managing three separate artworks within a single production run without cross-contamination or mislabeling
Decoration consistency — ensuring that spray coating and silkscreen printing held uniform standards across all 15,000 units
Packing execution — individual bagging protects surface finish during transit; palletization ensures safe, organized delivery at the filling end
In a scaling relationship, small orders are not throwaway exercises. They are the proving ground for whether a supplier can manage complexity — or only manage simplicity.
60ml Acrylic Dropper Bottle Program — Fitment and Component Coordination
The second early-stage product introduced a different class of challenge: the dropper system.
Key specifications:
| Parameter | Detail |
|---|---|
| Quantity | 10,000 pcs |
| Coating | Bright green spray coating |
| Printing | None (no silkscreen) |
| Dropper Components | Bright black dropper ring + matte black rubber bulb + glass pipette |
| Pre-Production Samples | 5 sets required |
| Packing | Bottles bagged; droppers packed separately in cartons; palletized |
Thread and torque consistency: a dropper ring that torques differently batch to batch creates inconsistent user experience and potential leakage
Pipette length alignment: if the glass pipette sits too short or too long within the bottle geometry, draw volume and control become unpredictable
Leak control during shipping: pressure differentials in transit can force fluid through improperly fitted assemblies
User experience consistency: if the dropper feel varies between reorders, end consumers notice — and retailers remember
Packing droppers separately from bottles was not an administrative formality. It was a deliberate protocol to prevent damage during transit, protect glass pipettes from breakage, and allow filling-line staff to assemble components in a controlled, organized manner.
This phase established the client’s foundational confidence in Jarsking’s ability to manage component complexity and deliver repeat-order stability.
Phase 2: Scaling into Jars — Where Cosmetic Quality Risk Becomes Visible
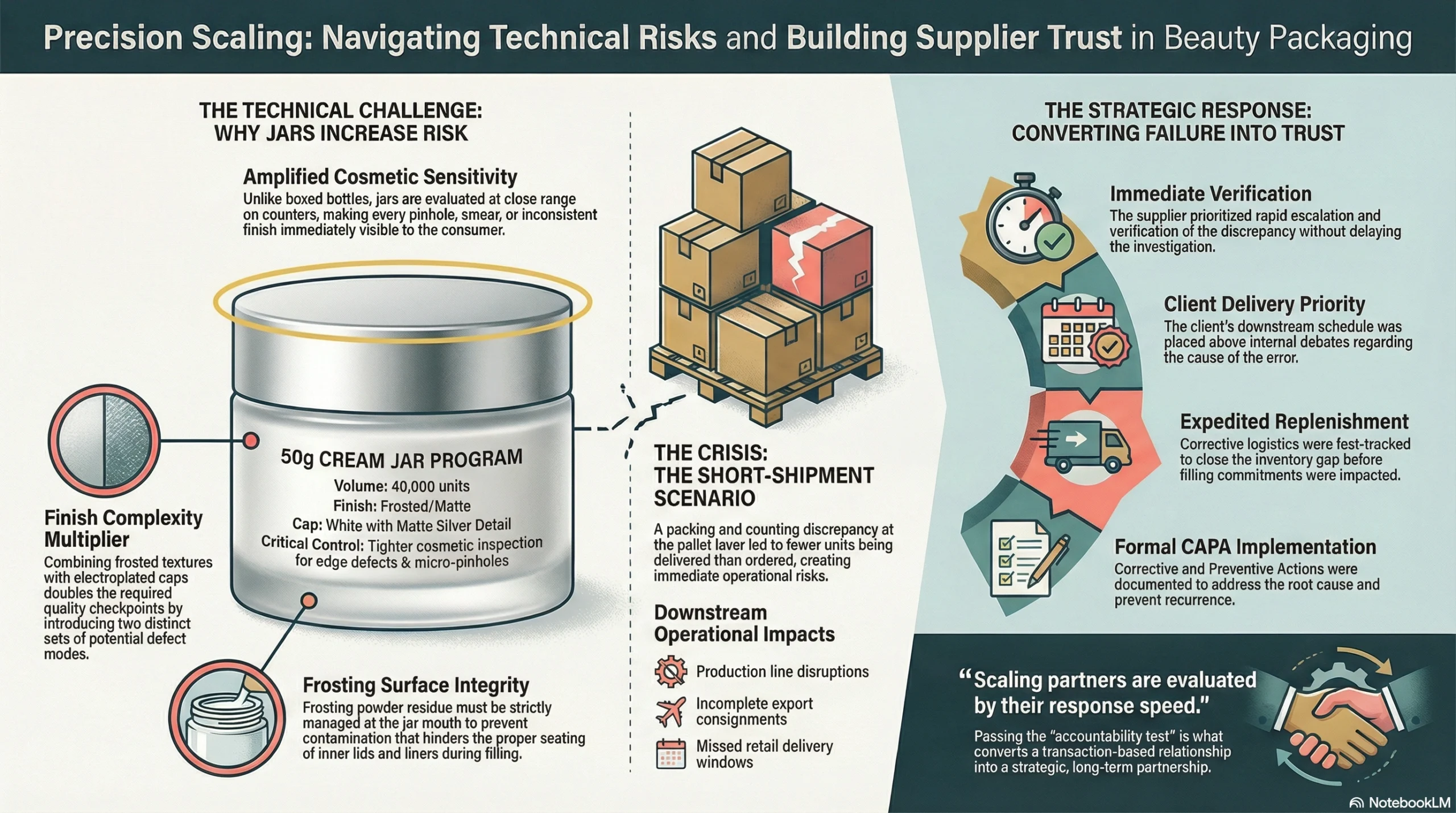
50g Cream Jar Program — Frosted Finish and Electroplated Cap
Scaling in beauty packaging is frequently won or lost in the jar category. The reason is architectural: jars amplify two risk types simultaneously.
First, cosmetic sensitivity — any surface defect on a jar is immediately visible to the consumer. Unlike a bottle hidden inside a box, a jar sits open on a shelf or a bathroom counter, where every pinhole, smear, or inconsistent finish is evaluated at close range.
Second, finish complexity — frosted textures and electroplated caps each introduce their own defect modes, and combining both in one product doubles the number of quality checkpoints required.
Key specifications:
| Parameter | Detail |
|---|---|
| Quantity | 40,000 pcs |
| Finish | Frosted / matte |
| Cap Assembly | White outer cap + electroplated matte silver detail |
| Critical Packing Note | Jars must be bagged; frosting powder must not remain at jar mouth |
| Cap Defect Controls | Edge defects monitored; surface pinholes and speckling controlled |
Controlling for this is not difficult — but it requires discipline. Jarsking built explicit process cleanliness controls and packing protocols to ensure the jar mouth arrived at the client’s filling line clean, every time.
The electroplated cap presented a separate challenge: edge defects and micro-pinholes in metallic plating are the cosmetic equivalent of a first impression gone wrong. Under consistent lighting — which is exactly how quality inspectors and end consumers evaluate beauty packaging — these defects stand out sharply against a smooth metallic surface.
This order required tighter cosmetic inspection standards and stricter acceptance criteria than anything the client had ordered before. It was also the order that raised the bar for every program that followed.
The Trust Moment: Accountability When It Matters Most
Every long-term supplier relationship contains a defining moment. For most brands, that moment arrives not when everything goes perfectly — but when something goes wrong.
During this partnership, a short-shipment scenario occurred. A packing and counting discrepancy at the pallet layer level resulted in the client receiving fewer units than ordered. For a business that supports multiple downstream brands and filling schedules, even a small count discrepancy creates immediate operational risk:
Production line disruption at the filling facility
Incomplete export consignments that can’t be released without full unit counts
Missed delivery windows to retail or distribution channels downstream
When this issue surfaced, Jarsking’s response followed a clear service logic:
Immediate escalation and verification — no delay in acknowledging and investigating the discrepancy
Client delivery priority over internal process — the client’s downstream schedule took precedence over internal debate about cause
Corrective replenishment under tight timelines — including expedited logistics to close the gap before the client’s filling commitments were impacted
Formal CAPA implementation — corrective and preventive actions documented and implemented to close the root cause
The reason this moment matters is straightforward: scaling partners do not evaluate their suppliers only by their perfect shipments. They evaluate suppliers by their response speed, their accountability, and their ability to protect the downstream schedule when reality doesn’t match the plan. That is the harder test — and passing it is what converts a transaction-based relationship into a strategic one.
Phase 3: The Breakthrough — Perfume Bottle System at Scale
A Platform Built for Repeatability, Not a Single Product
The client’s perfume program was never conceived as a one-time bottle order. From the start, the intent was a repeatable product family — one standardized bottle platform, multiple colorways, consistent appearance across all variants, stable fitment performance across every batch.
Compared with all earlier orders, the perfume system introduced structural complexity in three dimensions simultaneously:
Two-batch production planning — staged delivery to match the client’s inventory build and cash flow requirements
Tighter cosmetic expectations — coating defects that might be acceptable on a utility bottle become highly visible on a premium-positioned fragrance product
Functional performance requirements — vacuum sealing and adhesion must be proven by testing, not assumed from visual approval
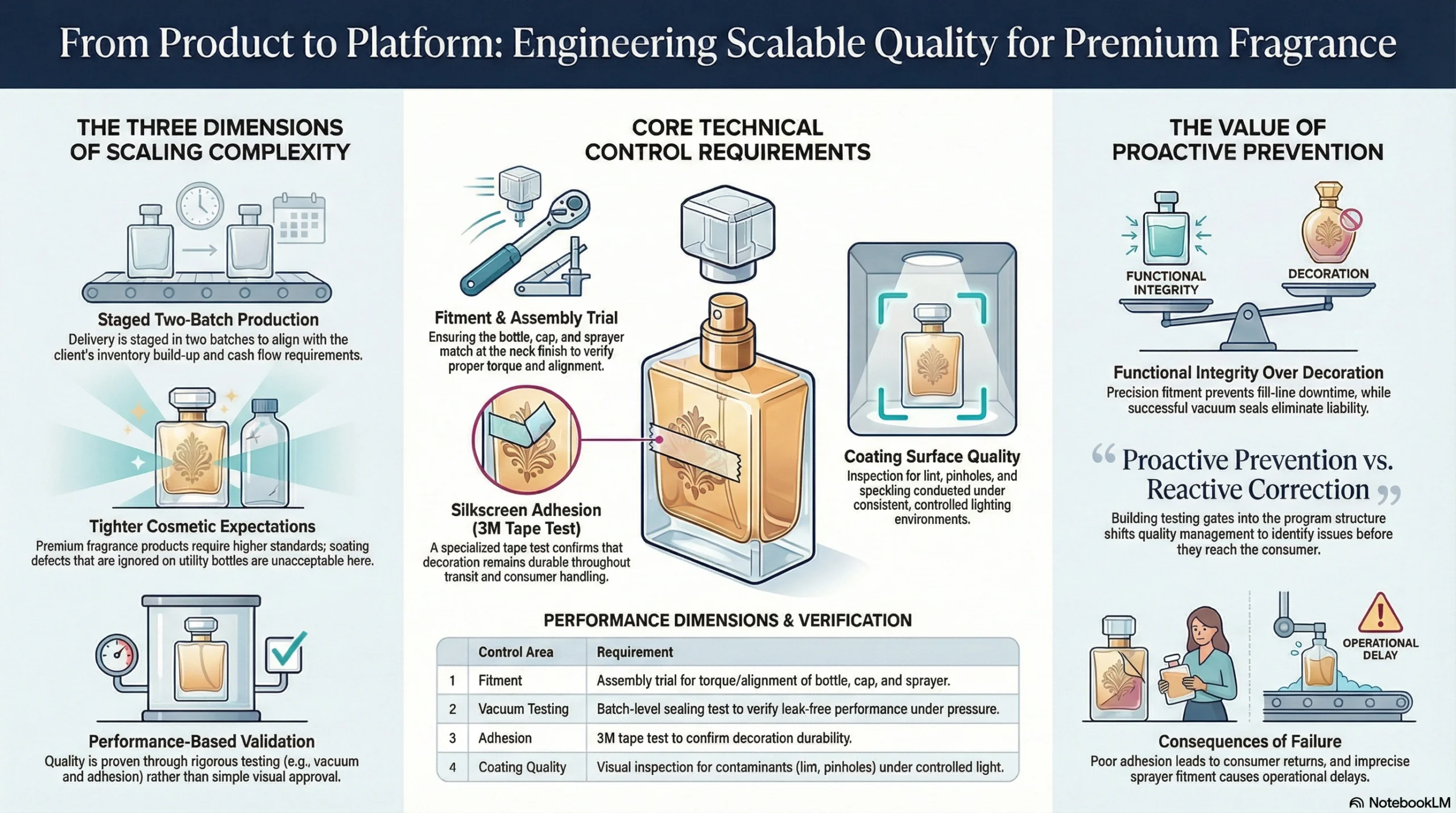
Core Technical Control Requirements for the 75ml Perfume Program
Before mass production was authorized, the program required verification across four performance dimensions:
| Control Area | Requirement |
|---|---|
| Fitment | Bottle, cap, and sprayer matched at neck finish; assembly trial to verify torque and alignment |
| Vacuum Testing | Batch-level sealing test to verify leak-free performance under pressure differential |
| Silkscreen Adhesion | 3M tape adhesion test to confirm decoration durability through transit and handling |
| Coating Surface Quality | Lint contamination, pinholes, and speckling inspected under consistent controlled lighting |
Production Data: Full Scope of the Partnership
Portfolio Milestones — Skincare + Fragrance
| Phase | Packaging Format | Quantity | Complexity Driver | Operational Focus |
|---|---|---|---|---|
| Phase 1 | 10ml roll-on bottle | 15,000 pcs | 3 designs + spray + silkscreen | PPS discipline, decoration repeatability, packing rules |
| Phase 1 | 60ml acrylic dropper bottle | 10,000 pcs | Dropper fitment + component packing | Component coordination, leak risk reduction |
| Phase 2 | 50g cream jar | 40,000 pcs | Frosting + electroplated cap | Cosmetic QC, finish cleanliness controls |
| Phase 3 | 75ml perfume system (bottle + sprayer sets) | 277,749 sets | Multi-colorways + testing gates | Fitment, vacuum test, 3M adhesion, staged delivery |
The perfume platform was executed in six distinct colorways across two production batches, allowing the client to service multiple SKUs within the same visual brand family.
| Colorway / Finish | Batch 1 Qty | Batch 2 Qty | Total Qty | Surface Process Notes |
|---|---|---|---|---|
| Pink | 16,178 | 25,633 | 41,811 | Semi-transparent coating + silkscreen |
| Rose | 25,980 | 37,160 | 63,140 | Semi-transparent coating + silkscreen |
| Black | 15,224 | 18,563 | 33,787 | Matte solid coating + white silkscreen |
| Tea | 14,905 | 20,473 | 35,378 | Semi-transparent coating + metallic finish element |
| Gold | 16,115 | 25,378 | 41,493 | Bright metallic effect + black silkscreen |
| Frosted | 25,780 | 36,360 | 62,140 | Frosted finish + silkscreen |
| Total Bottles | 114,182 | 163,567 | 277,749 |
| Sprayer Finish | Total Sets |
|---|---|
| Gold sprayer + collar | 181,822 |
| Silver sprayer + collar | 95,927 |
| Total Sprayer Sets | 277,749 |
The Execution Framework That Made It Repeatable
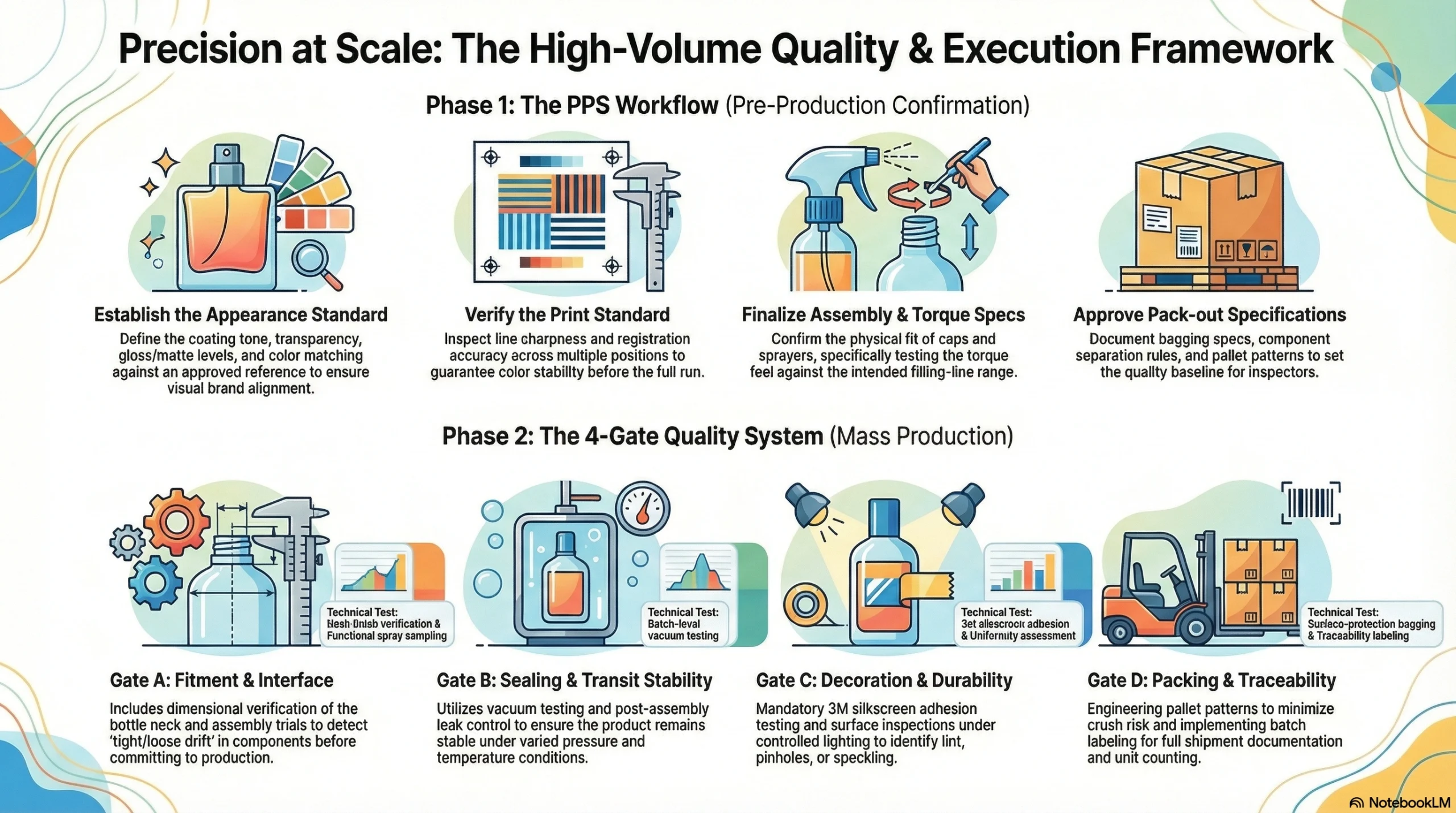
Pre-Production Sample (PPS) Workflow
Every order in this partnership — from the first 10,000-unit roll-on bottle to the 277,749-set perfume program — was gated by a formal pre-production sample confirmation before mass production commenced. The PPS workflow covers four confirmation areas:
Appearance standard: coating tone, transparency, gloss/matte level, and color matching to the approved reference
Print standard: line sharpness, registration accuracy, and color stability across multiple print positions
Assembly confirmation: cap fit, sprayer fit, torque feel at the filling-line torque range
Pack-out confirmation: bagging specification, component separation rules, and pallet pattern
PPS discipline does one essential thing: it removes ambiguity before production begins. When multiple stakeholders — procurement, marketing, and filling-line teams — have approved a tangible physical sample against documented standards, disputes about “what was agreed” disappear. Reorder consistency improves. And when volume scales, the approved PPS becomes the quality baseline that inspectors reference on every batch.
Quality Gate System for High-Volume Perfume Programs
For programs operating at the scale of this perfume project, Jarsking structured quality control across four formal gates — each targeting a distinct failure mode:
Gate A — Fitment Gate
Dimensional verification at the bottle neck finish and component interface
Assembly trial to detect “tight/loose drift” before full production commitment
Functional spray performance sampling from pre-production units
Gate B — Sealing Gate
Vacuum testing as a batch-level sampling protocol
Post-assembly leak control checks to ensure transit stability under varied pressure and temperature conditions
Gate C — Decoration Gate
3M silkscreen adhesion testing as a durability requirement, not an optional check
Coating surface inspection under controlled lighting for lint, pinholes, and speckling
Uniformity assessment across the full surface area of each colorway
Gate D — Packing Gate
Bagging requirements to protect finished coated surfaces from contact scratching
Palletization patterns engineered to minimize crush risk and support accurate unit counting
Carton labeling and batch traceability for full shipment documentation
Outcomes: What the Partnership Delivered
Operational Outcomes
The client transitioned from single-item standard orders into a fully integrated multi-category packaging system spanning skincare and fragrance
The perfume program was built and executed as a repeatable system — not a one-time production event
Testing gates (vacuum + adhesion) permanently shifted quality management from subjective visual approval to verified, documented performance
Commercial Growth Pattern
| Stage | Description |
|---|---|
| Early Stage | Small batches (10K–15K units) built communication rhythm and quality alignment |
| Breakthrough Stage | Large perfume program with six colorways, staged delivery, and formal testing protocol |
| Scale Stage | Annualized volumes exceeded 1,000,000 units on the same bottle platform |
| Forward Planning | Active discussions on doubling volume in the next planning cycle |
The client’s most important gain was not production capacity in isolation. It was control — across four dimensions that determine whether a beauty business can scale sustainably:
Control of cosmetic consistency across colorways and reorders
Control of fitment yield risk through engineering-level assembly verification
Control of repeatability through approved PPS references and documented standards
Control of delivery planning through two-batch execution aligned with inventory cycles
Practical Takeaways for Brands, Filling Factories, and Traders
If you are scaling from 10,000 to 100,000 units — or from 100,000 toward one million — the controls that protect your program are not complicated. But they must be built deliberately, and they must be built early.
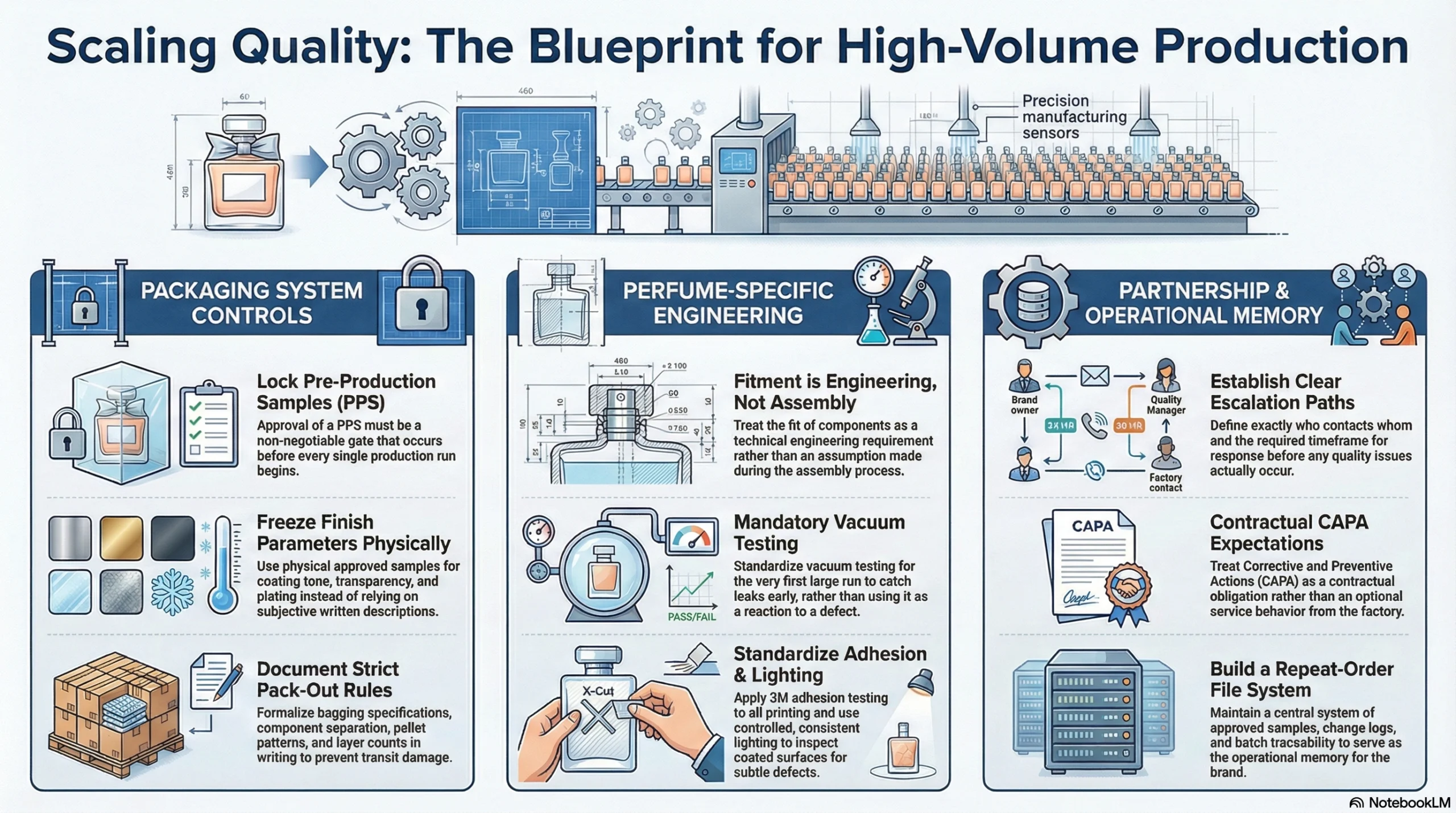
Packaging system controls:
Lock pre-production sample (PPS) approval as a non-negotiable gate before every production run
Freeze finish parameters — coating tone, transparency, plating reference — with physical approved samples, not written descriptions
Document pack-out rules in writing: bagging specification, component separation, pallet pattern, and layer count
Perfume-specific controls:
Treat fitment as an engineering requirement, not an assembly assumption
Require vacuum testing as a program standard from the first large run — not as an exception triggered by a defect event
Apply 3M adhesion testing to all silkscreen and printed decoration as a durability baseline
Inspect coated surfaces under controlled and consistent lighting to catch defects that daylight inspection routinely misses
Partnership controls:
Require a defined escalation path before a problem ever occurs — know exactly who contacts whom, and within what timeframe
Define corrective and preventive actions as a contractual expectation, not an optional service behavior
Build a repeat-order file system with approved samples, change logs, and batch traceability at its center — this is the operational memory that keeps quality consistent as your volume grows
Conclusion: Scale Is a System, Not a Number
The Eastern European beauty partner in this story didn’t reach one million units by placing bigger orders. They reached one million units by building a system — one that started with a 10,000-unit roll-on bottle and progressively added the controls, testing gates, component discipline, and partnership accountability that allow high volume to run smoothly.
The packaging supplier role in that journey was not passive. It required sample discipline, cosmetic quality leadership, honest escalation when things went wrong, and a continuous commitment to making the next order easier than the last.
If your business is in the early stages of building a repeatable beauty packaging program — or if you are already scaling and hitting quality or repeatability ceilings — Jarsking is ready to walk through your specific SKUs, volume targets, and category requirements. Contact Jarsking today to start building a packaging system that scales with you.
FAQs
The brand scaled by building a structured packaging system — not simply by ordering more. They started with small roll-on and dropper bottle batches to establish reliability, then progressively added cosmetic quality controls, formal testing gates, and disciplined pre-production sampling. By the time they launched their perfume program, they had the operational framework to handle 277,749+ sets across six colorways in two staged batches, leading to an annualized volume exceeding one million units.
A Pre-Production Sample (PPS) is a physical, approved reference unit produced before mass manufacturing begins. It locks in appearance standards (coating, transparency, gloss level), print quality, assembly fit, and packing specifications. For scaling brands, PPS approval removes ambiguity, prevents disputes, and ensures that every reorder — at any volume — is benchmarked against a documented, agreed standard rather than memory or written descriptions alone.
Three critical tests are recommended for perfume programs at scale. First, vacuum testing verifies that the bottle and sprayer assembly is leak-free under pressure differentials encountered during transit. Second, 3M adhesion testing confirms that silkscreen and printed decoration will not peel or degrade through normal handling. Third, fitment verification ensures that the bottle neck, sprayer, and cap interface correctly with consistent torque — preventing fill-line downtime and user experience failures.
Jars amplify two risk types simultaneously. First, cosmetic sensitivity — any surface defect is immediately visible because jars are displayed openly on shelves and countertops, where consumers inspect them closely. Second, finish complexity — processes like frosting and electroplating introduce additional defect modes such as residual powder at the jar mouth, edge defects on plated caps, and surface pinholes. These risks require tighter cosmetic inspection standards and stricter process cleanliness controls than most bottle formats.
A short-shipment or count discrepancy should trigger an immediate four-step response from the supplier: acknowledgment and verification without delay, prioritization of the client’s downstream delivery schedule, execution of a corrective replenishment plan under tight timelines, and formal implementation of corrective and preventive actions (CAPA) to eliminate the root cause. Brands should establish a clear escalation path and define CAPA expectations as a contractual requirement — before a problem ever occurs.
Building multiple SKUs on a single standardized bottle platform delivers three key advantages. It reduces tooling and mold costs because only surface finishing — coating color, silkscreen design — varies between variants, not the bottle geometry itself. It creates a consistent brand silhouette across the full fragrance range, strengthening visual identity on shelf and in e-commerce imagery. And it simplifies reorder management, because fitment specifications, testing protocols, and PPS references remain consistent across every colorway.