If you’re preparing to sell cosmetics in U.S. supermarkets, you’ve already discovered that getting your packaging right is far more complex than picking an attractive bottle and printing a label. U.S. cosmetic packaging standards for supermarkets sit at the intersection of federal regulation, retail operations, and consumer trust—and failing on any one of them can delay or derail your retail launch entirely.
Here’s a direct answer to the core question: supermarket-ready cosmetic packaging in the United States must satisfy FDA cosmetic labeling rules, MoCRA obligations, Fair Packaging and Labeling Act disclosures, FTC claim guidance, and GS1 US barcode standards—while also being shelf-durable, easy to scan, and visually compelling enough to earn a second look.
This guide breaks each of those requirements into practical packaging decisions. You’ll learn exactly what information must appear on your labels, how MoCRA is changing what brands need to disclose, what barcodes you need and where to place them, and how to make sustainability claims without triggering greenwashing liability. Along the way, you’ll see how an experienced packaging manufacturer like Jarsking can help you navigate every step—turning compliance obligations into packaging assets rather than roadblocks.
Why Supermarkets Have Different Packaging Expectations Than Online Beauty Sales
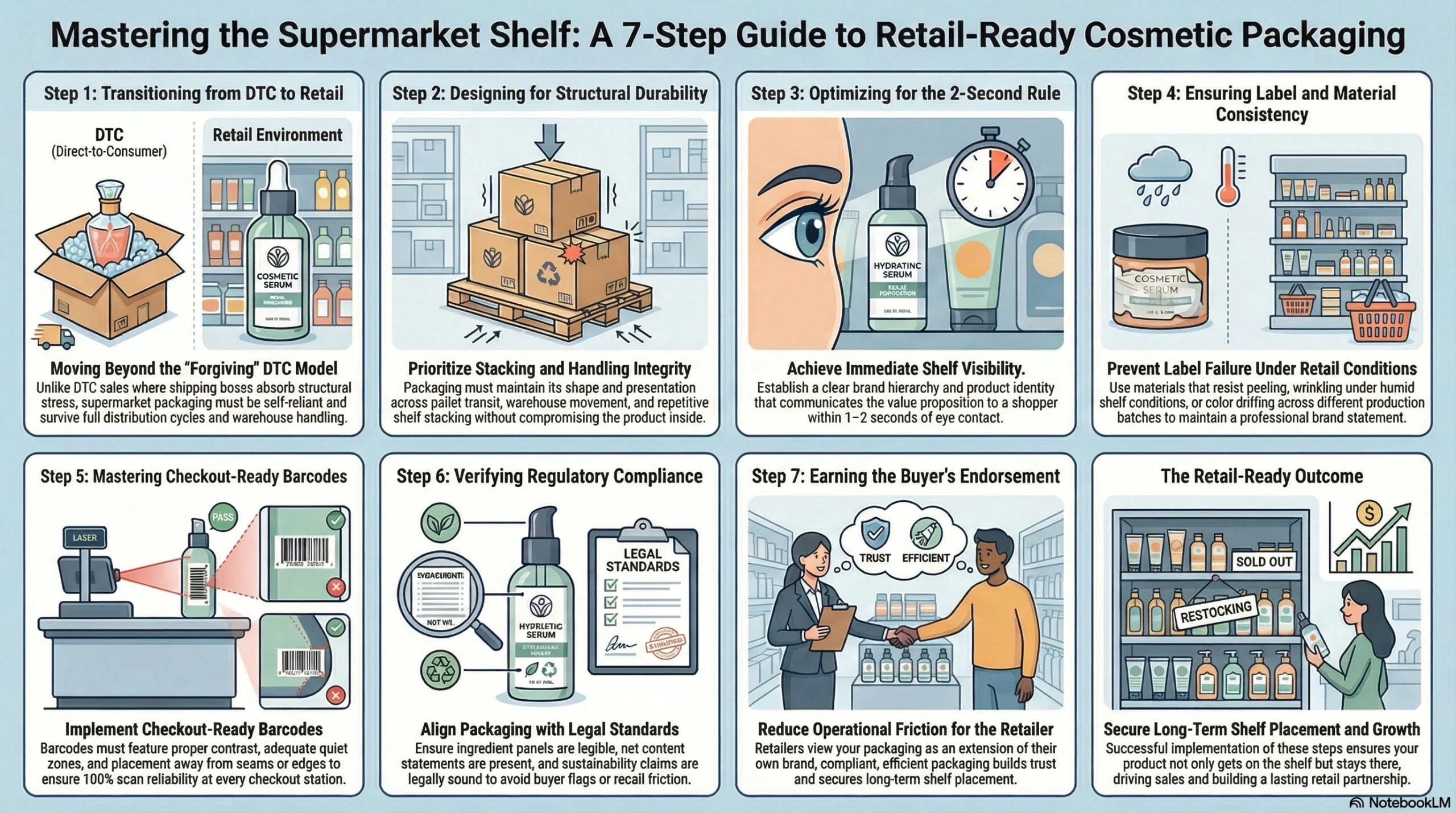
Selling cosmetics through a DTC website is forgiving. Customers search for your brand by name, product photos can be updated instantly, and shipping boxes absorb a lot of structural imperfection. Supermarket retail is an entirely different operating environment—and your packaging must perform across three simultaneous dimensions before a single unit sells.
Supermarket Packaging Must Perform on Shelf, in Transit, and at Checkout
Retail buyers don’t just evaluate how a product looks in a mockup. They evaluate whether it can survive full distribution cycles, stack cleanly on-shelf, hold its label presentation under fluorescent lighting, and scan accurately at the point of sale. A jar with a gorgeous label that wrinkles after two weeks on a humid shelf, or a tube whose barcode sits too close to the bottom seam, will generate costly returns and stocking complaints that damage your buyer relationship.
Specifically, supermarket-ready cosmetic packaging must deliver:
Shelf visibility: Clear product identity and brand hierarchy that communicates within 1–2 seconds
Stacking and handling durability: Structural integrity across warehouse handling, pallet transit, and shelf stocking
Inventory traceability: Barcodes that scan reliably across every unit and every checkout station
Consistent label presentation: No peeling, wrinkling, or color drift across production batches
Barcode readability: Proper placement, contrast, quiet zones, and print quality
Retail Buyers Care About Compliance, Efficiency, and Shopper Clarity
Supermarket category buyers are gatekeepers. Before approving a new cosmetic line for shelf placement, they want confidence that the brand won’t create compliance liability, recall friction, or shopper confusion. They look for packaging that reduces their operational friction in stocking, scanning, and managing inventory—not packaging that creates it.
From a retailer’s perspective, every cosmetic package on their shelf is also a brand statement they’re implicitly endorsing. If your ingredient panel is hard to read, your net content statement is missing, or your sustainability claims look legally risky, a well-informed buyer will flag those problems before your product ever reaches the shelf. That’s why retail-ready beauty packaging and regulatory compliance are not separate tasks—they’re the same task, approached from two directions.
The Core U.S. Rules That Shape Cosmetic Packaging
Understanding the regulatory framework first makes every subsequent packaging decision easier. There are four primary legal frameworks every brand must know.
FDA Cosmetic Labeling Basics
The FDA regulates cosmetics sold in the U.S. under the Federal Food, Drug, and Cosmetic Act (FD&C Act) and the Fair Packaging and Labeling Act (FPLA). Labeling requirements are codified at 21 CFR 701 and 740, and they apply to all cosmetics marketed domestically, whether manufactured in the U.S. or imported.
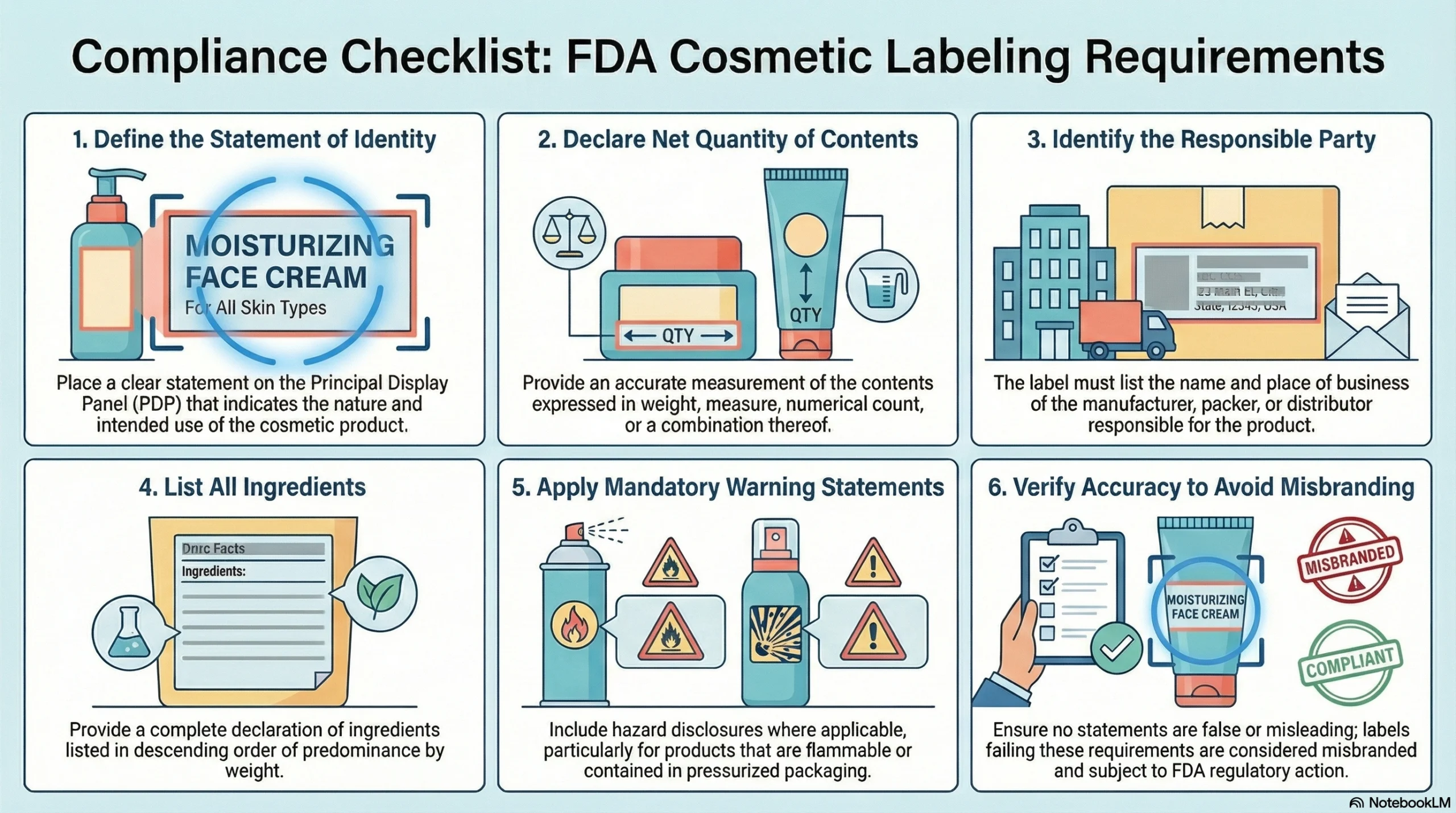
The FDA requires that every retail cosmetic package carry:
An identity statement on the principal display panel indicating the nature and use of the product
An accurate net quantity of contents expressed in weight, measure, or numerical count
The name and place of business of the manufacturer, packer, or distributor
A complete ingredient declaration in descending order of predominance
Required warning statements where applicable, including hazard disclosures for flammable or pressurized products
Cosmetics bearing false or misleading label statements, or labels that fail to meet these requirements, can be considered misbranded and subject to regulatory action.
MoCRA and Why It Matters for Packaging Teams
The Modernization of Cosmetics Regulation Act of 2022 (MoCRA) is the most significant update to U.S. cosmetic regulation in over 80 years. For packaging teams, MoCRA introduces obligations that go well beyond label text—they reshape how brands think about traceability, safety data, and consumer communication.
The key MoCRA packaging implications include:
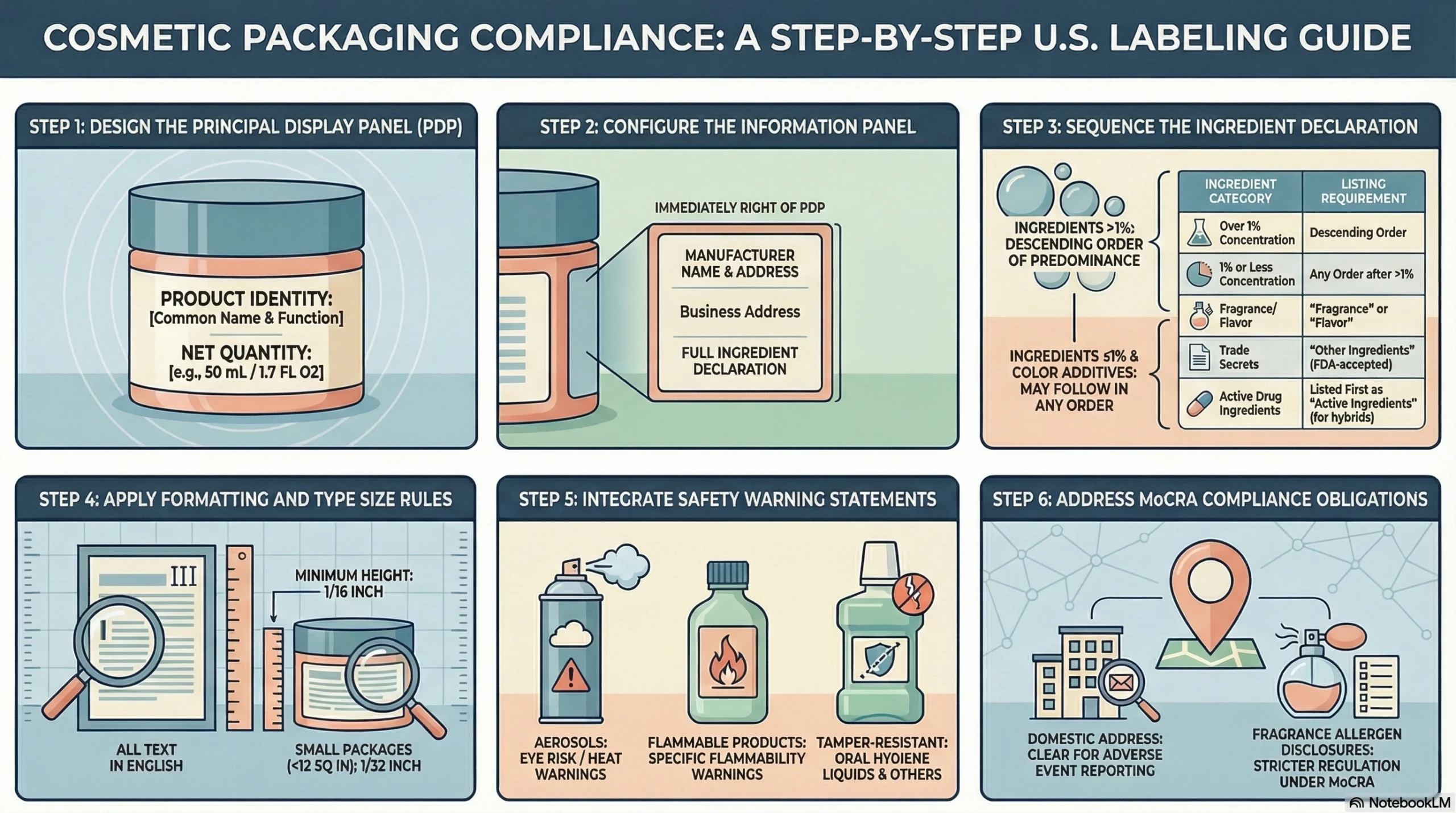
Domestic contact information on labels: As of December 29, 2024, all cosmetic labels must include a domestic address, phone number, or electronic contact information so consumers and regulators can report adverse events
Adverse event reporting: Responsible persons must report serious adverse events to the FDA within 15 business days and must include a copy of the product label in each report
Record-keeping: Adverse event records must be maintained for six years (three years for small businesses)
Product listing: All cosmetics must be listed with the FDA, with ingredient lists that must exactly match what appears on the label
Fragrance allergen disclosure: MoCRA directs the FDA to determine which fragrance allergens must be named on labels individually rather than grouped under “fragrance”
MoCRA adds major operational pressure to cosmetic packaging because labels now sit closer to post-market safety and adverse-event workflows. Packaging decisions made today will affect your ability to comply with MoCRA obligations for years to come—so building contact information placement and label accuracy into your packaging design process from the start is no longer optional.
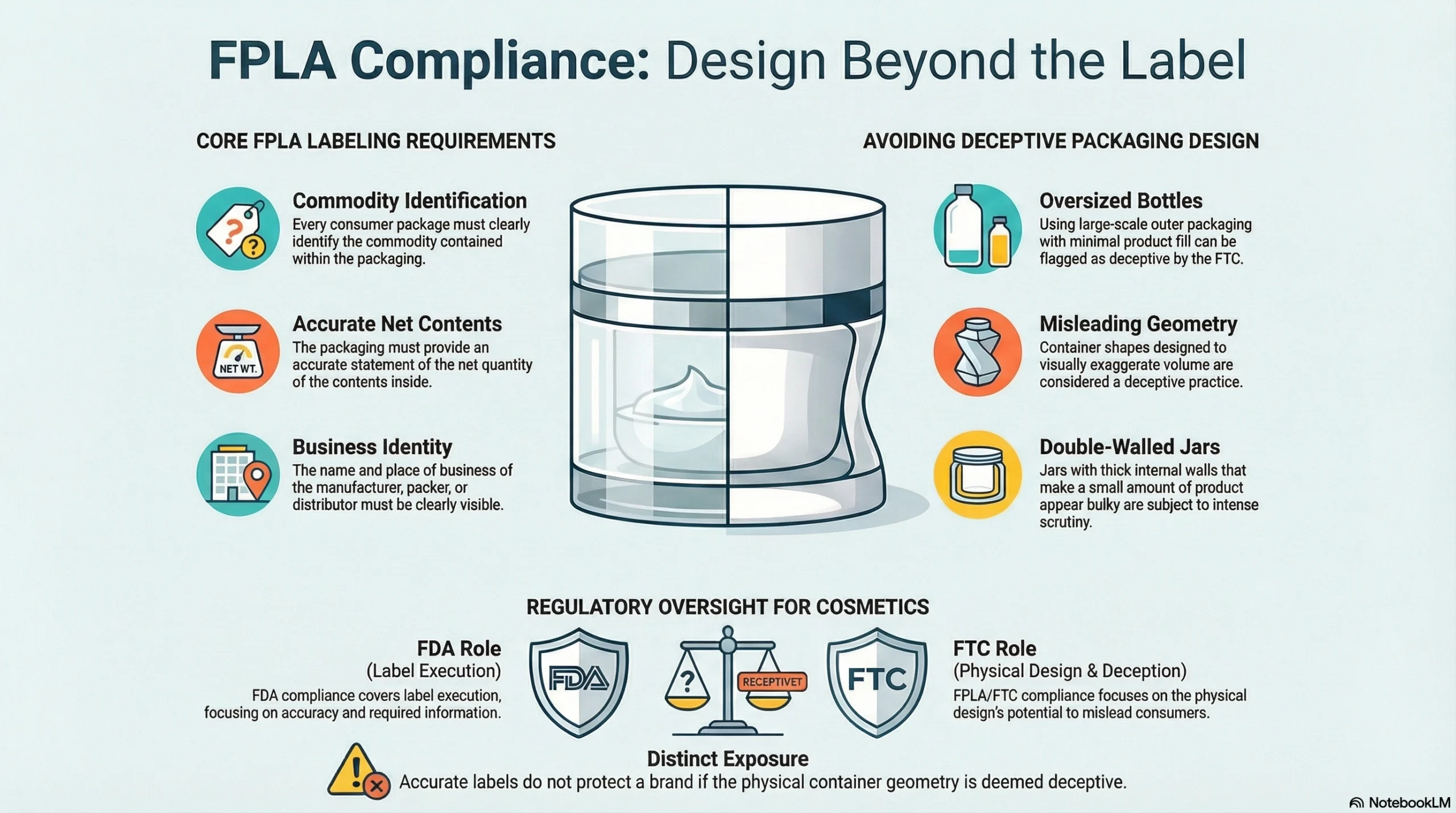
FTC and Fair Packaging and Labeling Act Basics
The Fair Packaging and Labeling Act, enforced by the FTC, requires that consumer packages identify the commodity, accurately state the net contents, and provide the manufacturer or distributor’s name and place of business. The broader purpose is to prevent deceptive packaging—meaning package sizes, shapes, and structural choices can themselves become regulatory issues if they create misleading impressions about quantity or value.
For cosmetic brands, FPLA compliance is usually satisfied by accurate FDA label execution. But where FPLA adds distinct exposure is in packaging design itself: oversized bottles with minimal fill, misleading container geometry, or deceptive double-walled jars that exaggerate perceived product volume can all attract FTC scrutiny.
NIST and Measurement Presentation Standards
NIST provides guidelines that inform how quantities must be expressed on consumer packages. Net quantity statements for solid or semi-solid cosmetics must be expressed in avoirdupois pounds and ounces, while liquid cosmetics must use U.S. gallon, quart, pint, or fluid ounce measurements. Brands entering from international markets—where metric labeling is standard—should also include metric equivalents, as dual-unit labeling is generally expected for mainstream U.S. retail.
What Information Must Appear on Cosmetic Packaging for U.S. Supermarkets
Regulations translate into specific label placement decisions. Here is how the rules map to the physical surface of your packaging.
What Belongs on the Principal Display Panel
The principal display panel (PDP) is the portion of the label most likely to be seen by consumers at the point of purchase. It must carry:
Product identity statement: The common, descriptive, or fanciful name that clearly indicates what the product is and what it does
Net quantity of contents: Placed in the bottom area of the PDP, parallel to the base, in a type size commensurate with container size
The PDP must present this information prominently and conspicuously, so consumers can understand it under normal retail conditions.
What Belongs on the Information Panel
The information panel—typically the surface immediately to the right of the PDP—carries the detailed disclosure content required by the FDA and FPLA:
Name and place of business: Manufacturer, packer, or distributor, with qualifying phrases like “Manufactured for” or “Distributed by” when the named entity is not the manufacturer
Domestic address: Required to be present, especially now under MoCRA adverse event contact obligations
Ingredient declaration: Full ingredient list in descending order of predominance
All label text required by regulation must be in English and must be placed with sufficient prominence and conspicuousness that consumers can read it under normal purchase conditions.
Ingredient Declaration Rules Brands Often Overlook
Ingredient declaration follows specific rules that brands frequently get wrong in early packaging iterations:
Ingredients must be listed in descending order of predominance
Ingredients present at 1% or less may be listed in any order, as long as they follow higher-concentration ingredients
Color additives may be listed in any order after all other ingredients
Fragrance and flavor may be listed as “Fragrance” or “Flavor” rather than by individual components—though MoCRA is narrowing this exception for known allergens
Ingredients accepted by the FDA as trade secrets may be listed as “Other Ingredients”
For cosmetics that are also drugs, active drug ingredients must be listed first as “Active Ingredients”
The FDA Cosmetics Labeling Guide is especially useful here because it gives practical rules for ingredient presentation, type size, and small-pack exceptions. Minimum type height for ingredient declarations is 1/16 of an inch; for packages with total surface area less than 12 square inches, 1/32 of an inch is permitted.
Warning Statements and Special Cases
Certain cosmetics require specific warnings by regulation:
Aerosol/pressurized containers: Must bear a multi-part warning covering eye spray risk, pressure, heat storage, and child safety
Flammable products: Must be labeled accordingly under 21 CFR 740.1
Liquid oral hygiene products and vaginal cosmetics: Must be packaged in tamper-resistant packaging when sold at retail
Omitting required warnings—even inadvertently—creates misbranding risk and potential liability. For brands managing multiple SKUs, building a warning-statement review into the packaging development checklist is essential.
Small Containers, Travel Sizes, and Decorative Packs Need Smarter Label Planning
Mini formats, travel sizes, and luxury decorative packaging are high-growth segments in supermarket beauty. They also create concentrated compliance pressure because limited surface area must still carry all required disclosures.
Why Small Cosmetic Packaging Creates Compliance Pressure
FDA Flexibility for Small or Special Containers
The FDA provides limited but important flexibility for small packages:
Packages with less than 12 square inches of total surface area may use 1/32-inch minimum type height for ingredient declarations
Off-package ingredient labeling is permitted under 21 CFR 701.3(i) if the cosmetic is held in tightly compartmented trays or racks, is not enclosed in a folding carton, and has less than 12 square inches of package surface area
These exceptions allow brands to use display cards, hang tags, or counter displays that carry the full ingredient declaration when the primary container cannot accommodate it
Packaging Design Strategies for Small-Format Compliance
The most effective approach for small cosmetic packaging is to plan label space before finalizing structural design—not after:
Secondary cartons: Outer cartons carry the full ingredient declaration and detailed disclosures while the inner container carries the PDP essentials
Wrap-around labels: Enable maximum text coverage on cylindrical formats like lip balm tubes, roll-ons, and narrow serums
Fold-out labels: Accordion-style inserts attached to the container allow full disclosure without disrupting premium aesthetics
Display cards or backer cards: Used in retail display systems where the card itself carries required text for very small containers
The key is briefing your packaging manufacturer early with your full label content requirements, so structural design accommodates compliance from the first prototype.
Barcode and Retail-Ready Packaging Requirements for Supermarkets
Why Cosmetics Need Barcodes for Supermarket Distribution
Supermarkets run on barcode infrastructure. Every SKU that enters a retail distribution system needs a scannable, globally unique identifier for:
Inventory accuracy: Retailers track stock levels, reorder points, and shrinkage by barcode
POS scanning: Checkout registers retrieve pricing, promotions, and product data via barcode scan
Retail onboarding: Category buyers and EDI systems use GTINs to set up product listings
Traceability and recall management: In the event of a product recall, barcodes allow retailers to identify and pull specific lots quickly
GS1 US notes that barcodes are essential for retail inventory management, supply-chain visibility, and product recalls. Online marketplaces like Amazon and major grocery retailers use the GS1 database to verify the authenticity of your cosmetic products, which means barcodes purchased from third-party resellers—who may recycle or reassign numbers—will fail validation.
Barcode Type and Placement Basics
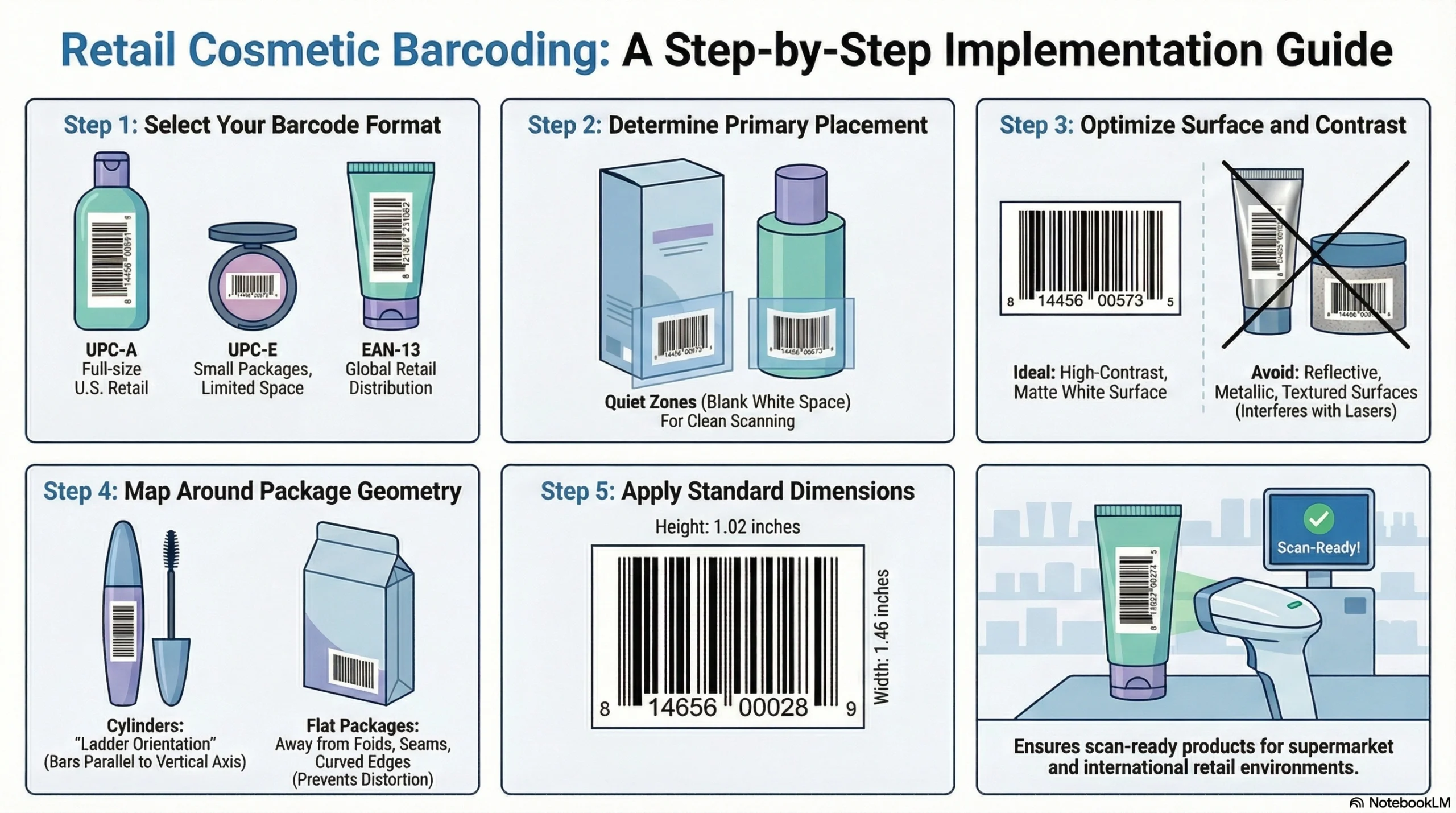
For most retail cosmetics, the relevant barcode formats are:
UPC-A: The standard 12-digit barcode used for full-size cosmetic packages sold through U.S. supermarkets
UPC-E: A compressed 6-digit version permitted for smaller packages where space constraints make UPC-A impractical
EAN-13: The 13-digit international standard, accepted at most major U.S. retailers and required for brands selling across both U.S. and international markets
Regarding placement, GS1 US recommends putting retail barcodes in the lower right-hand section of the back of the package and preserving adequate white space—called “quiet zones”—for clean scans. Additional placement best practices include:
Avoid placing barcodes near folds, seams, or curved edges where substrate distortion can cause scan failure
Maintain high contrast between barcode bars and background—black on white is ideal; avoid printing on reflective, metallic, or textured surfaces without testing
Use smooth, non-gloss print surfaces wherever possible
For cylindrical containers, orient barcodes so bars run parallel to the vertical axis of the container (“ladder orientation”)
Standard recommended barcode size for retail cosmetics is approximately 1.46 inches wide by 1.02 inches tall.
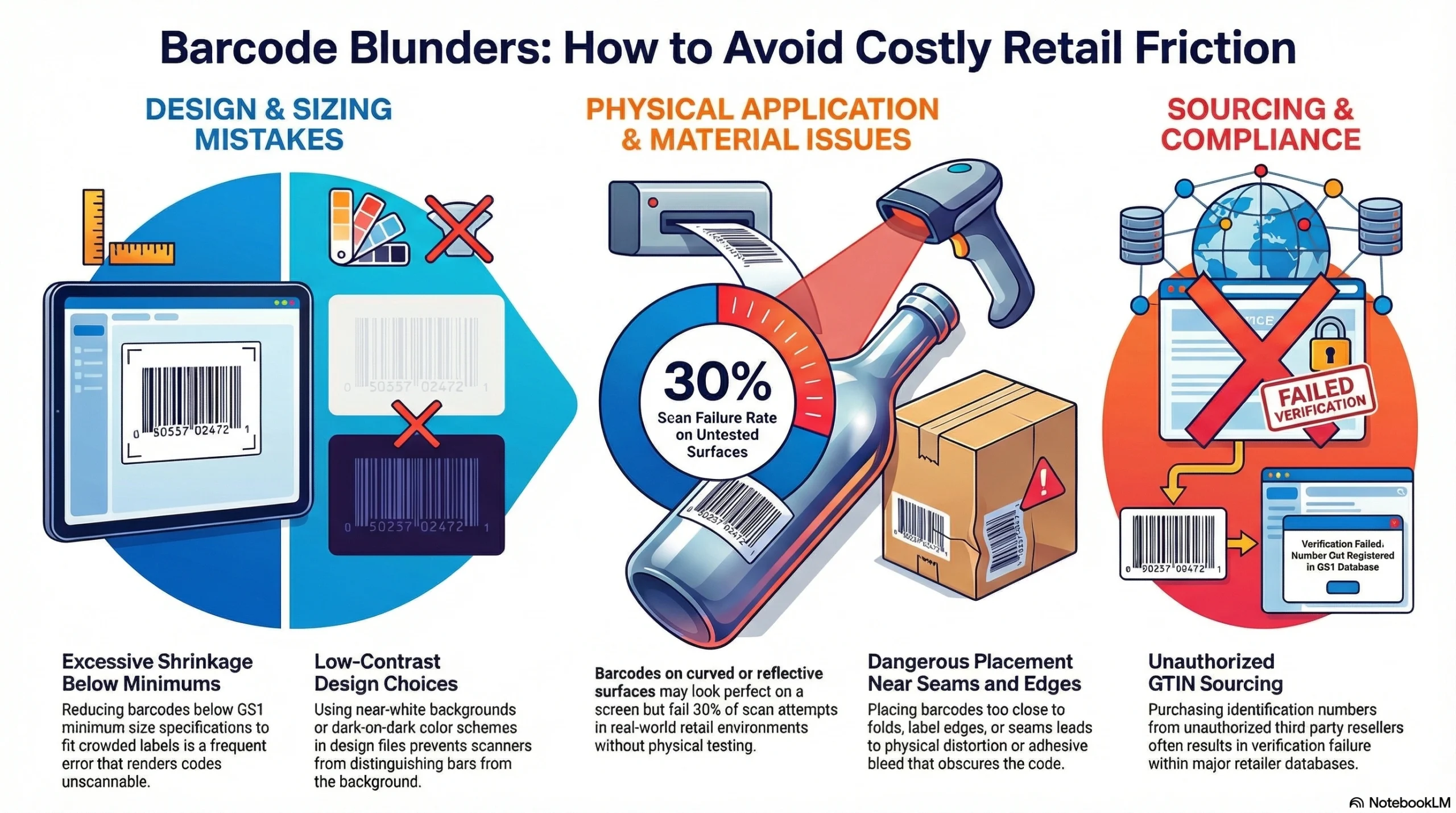
Common Barcode Mistakes That Lead to Retail Friction
Even brands that understand the rules make operational barcode errors that create expensive problems:
Shrinking barcodes below minimum scan size to fit a crowded label layout—always verify against GS1 minimum size specifications before printing
Printing on curved or reflective surfaces without physical scan testing—a code that looks correct on screen may fail 30% of scan attempts in real retail conditions
Placing barcodes too close to seams, folds, or label edges where distortion or adhesive bleed can obscure bars
Poor print contrast from design files that use near-white backgrounds or dark-on-dark color choices
Purchasing GTINs from unauthorized third-party resellers whose numbers fail GS1 database verification at major retailers
Packaging Claims, Sustainability Messaging, and Greenwashing Risk
Why Packaging Claims Matter as Much as the Package Itself
Supermarket shoppers make purchase decisions in seconds, often based on a single phrase or visual cue on the front of the package. This means claims like “sustainable,” “recyclable,” “refillable,” “clean beauty,” “made in USA,” and “eco-friendly” carry commercial weight—and that commercial weight is precisely what attracts FTC scrutiny.
The FTC’s Green Guides establish that marketers cannot make broad, unqualified environmental claims about their products. The overall impression of packaging—not just the specific text used—determines whether a claim is legally defensible.
FTC Claim Guidance Brands Should Keep in Mind
Key principles from FTC Green Guides guidance that apply directly to cosmetic packaging include:
Vague terms need substantiation: “Eco-friendly,” “green,” and “clean beauty” are considered broad environmental claims that require real, documentable backing—not just brand intention
Packaging visuals can imply claims: Using green color schemes, leaf imagery, or natural textures can create the impression of environmental benefit even when no explicit claim is made
Recyclable claims require honest qualifications: A package is only “recyclable” if a substantial majority of consumers have access to facilities that will actually accept and recycle it
Single-attribute claims should not imply whole-product benefits: Saying a package uses “30% recycled content” is not the same as calling the entire product sustainable—and conflating the two can create liability
The FTC has signaled that its forthcoming Green Guides update will provide more clarity and potentially increase scrutiny for brands that continue to use vague sustainability labels without substantiation.
"Made in USA" and Origin Claims on Cosmetic Packaging
Origin claims are a particularly nuanced area for cosmetic brands, especially those who manufacture components internationally and assemble or fill domestically:
“Made in USA” requires that the product be all, or virtually all, made in the United States. This includes components and materials, not just assembly.
Flags, icons, and phrasing can imply a “Made in USA” claim even when no explicit statement is made
Qualified claims are safer when narrowly true: “Filled in the USA” or “Packaged in the USA from imported components” is legally cleaner than an unqualified origin claim
FTC guidance makes clear that the overall impression of packaging can create origin claims, and that “Packaging Made in USA” is not the same as saying the product itself is made in the U.S. Always review origin claim language with legal counsel before printing.
How to Talk About Sustainable Packaging Credibly
Sustainable packaging is increasingly important to supermarket buyers and their shoppers—and it’s possible to communicate sustainability responsibly without legal risk:
PCR (post-consumer recycled) content: Specify the exact percentage (e.g., “Made with 50% PCR materials”) rather than using generic “recycled” language
Refill systems: If your packaging supports a verified refill program, the mechanism and availability should be clearly described
Recyclable glass or mono-material structures: These can be marketed accurately when the infrastructure to recycle them actually exists in the markets where you sell
Third-party certifications: Programs like How2Recycle provide verified, industry-recognized recyclability claims that carry more weight than unsubstantiated marketing language
Life Cycle Assessments (LCAs): For brands making comprehensive sustainability claims, LCAs provide documented evidence of environmental performance across the full product lifecycle
Common Cosmetic Packaging Mistakes That Delay Supermarket Entry
Understanding what can go wrong is as valuable as knowing what to do right. These are the most common packaging mistakes that derail supermarket entry for beauty brands.
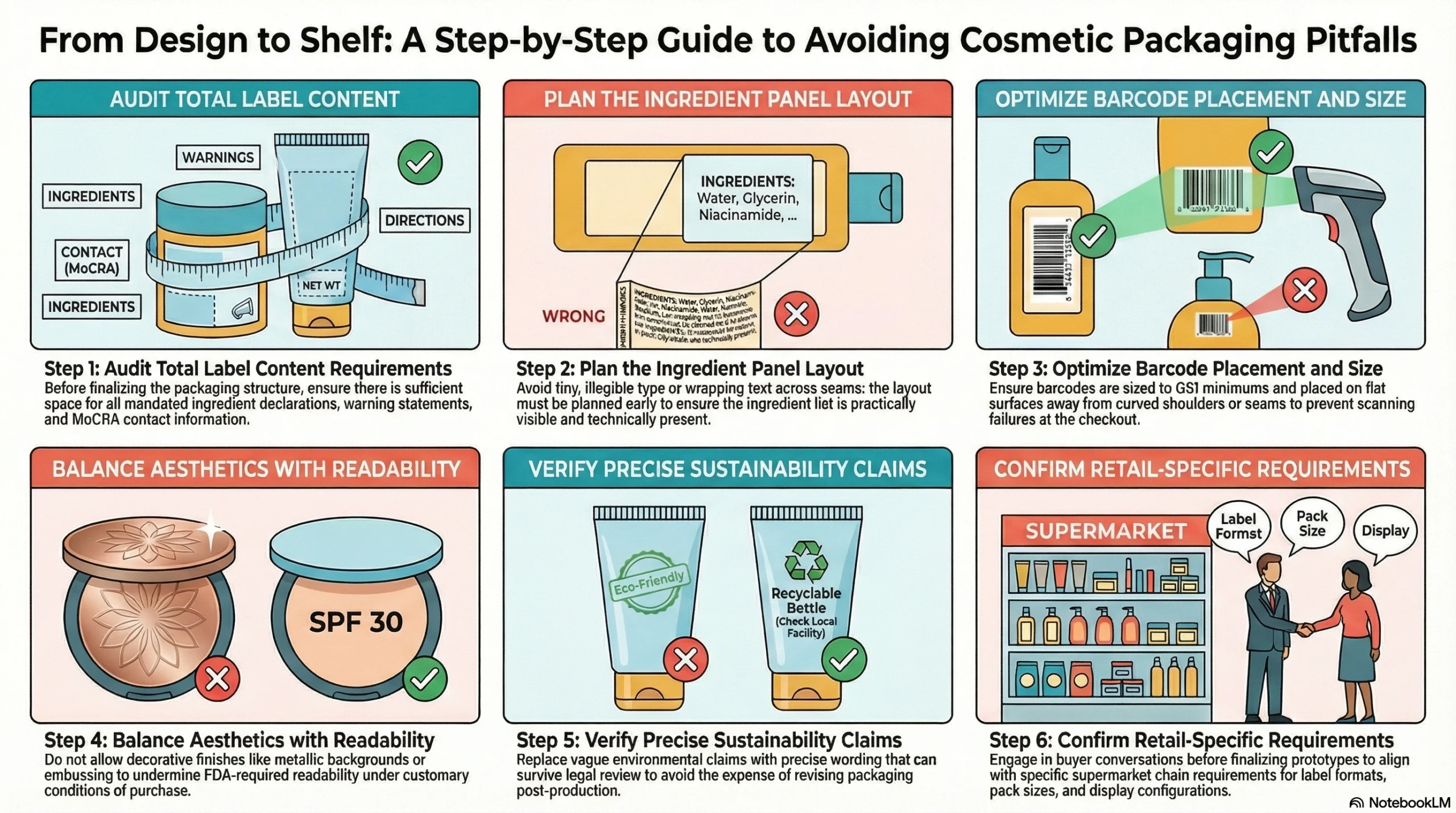
Missing or crowded label content: Brands often finalize packaging structure before auditing total label content requirements—then discover they don’t have enough space for ingredient declarations, warning statements, and MoCRA contact information
Poor ingredient panel planning: Rushing ingredient layout into the final stages of design leads to type that’s too small to read, ingredient lists that wrap awkwardly across seams, or declarations that are technically present but practically invisible
Barcode placement that fails at checkout: A barcode printed on a curved shoulder, placed over a seam, or sized below GS1 minimums will fail scan tests—often discovered only after production runs are complete
Decorative packaging that sacrifices readability: Embossed surfaces, dark label backgrounds, and metallic finishes can all undermine the readability requirements that FDA requires under customary conditions of purchase
Sustainability claims without precise wording: Vague environmental claims made under time pressure frequently don’t survive legal review—and revising packaging claims post-production is expensive
Designing packaging before confirming retail requirements: Each supermarket chain may have specific requirements regarding label formats, barcode types, pack sizes, and display configurations—skipping buyer conversations before finalizing packaging structure wastes prototyping cycles
A Practical Packaging Checklist for Beauty Brands Selling Through U.S. Supermarkets
Use this checklist during your packaging development process to validate readiness before committing to production.
Compliance Checklist
Product identity statement present on the principal display panel
Net quantity of contents stated in correct units, placed in the bottom area of the PDP
Manufacturer/distributor name and domestic address present on the information panel
Ingredient declaration in descending order of predominance, with correct handling of color additives, fragrance, and 1% threshold
All required warning statements reviewed and included (aerosol, flammable, tamper-resistant packaging where applicable)
MoCRA adverse event contact information (domestic address, phone number, or electronic contact) confirmed on label
Label content matches the ingredient list submitted to the FDA for product listing
Fragrance allergen disclosure reviewed in light of MoCRA requirements
Type size verified to meet minimum FDA requirements for the package surface area
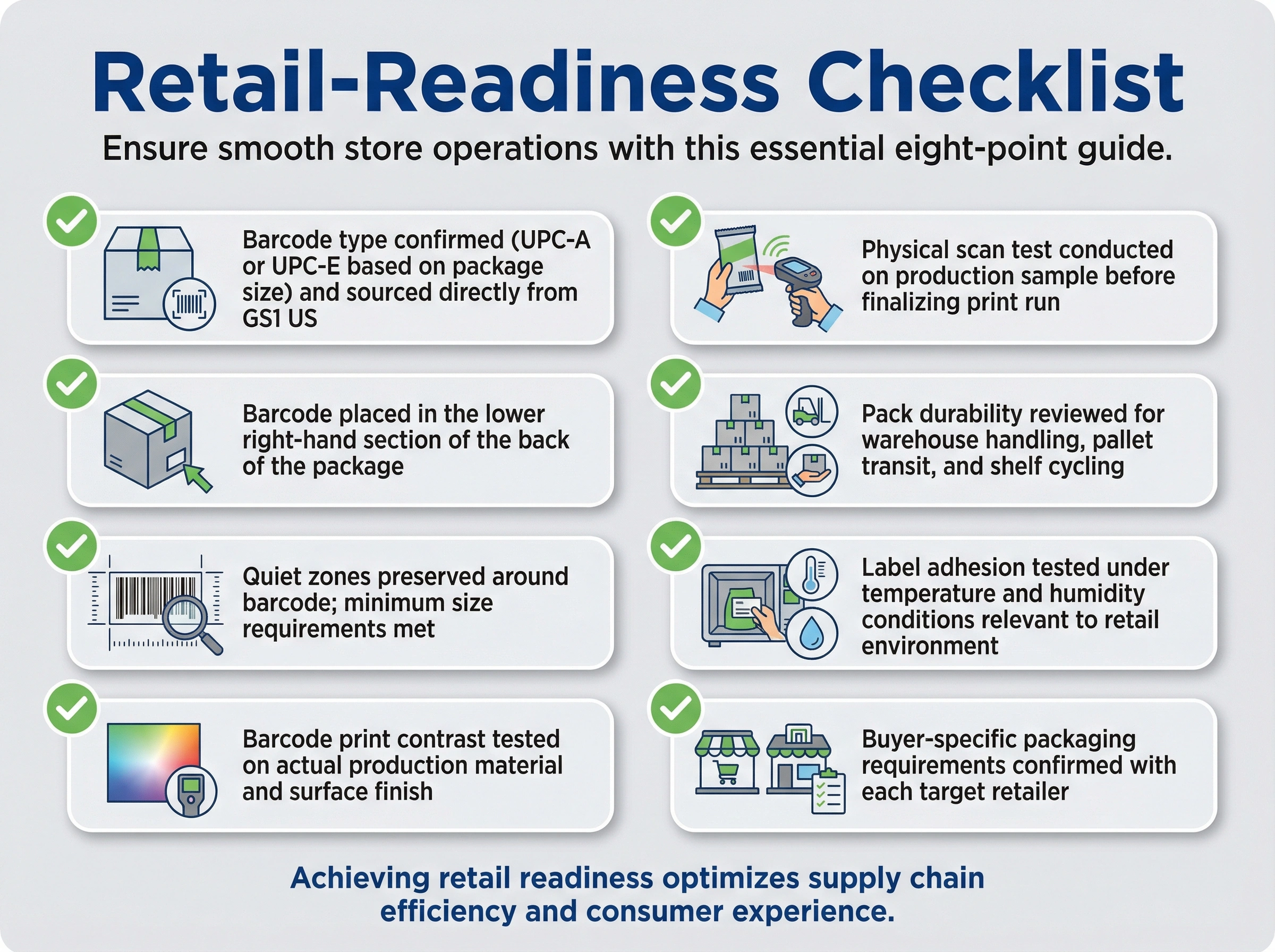
Retail-Readiness Checklist
Barcode type confirmed (UPC-A or UPC-E based on package size) and sourced directly from GS1 US
Barcode placed in the lower right-hand section of the back of the package
Quiet zones preserved around barcode; minimum size requirements met
Barcode print contrast tested on actual production material and surface finish
Physical scan test conducted on production sample before finalizing print run
Pack durability reviewed for warehouse handling, pallet transit, and shelf cycling
Label adhesion tested under temperature and humidity conditions relevant to retail environment
Buyer-specific packaging requirements confirmed with each target retailer
Sustainability and Claims Checklist
All environmental claims reviewed against FTC Green Guides standards
PCR or recycled content claims state specific percentages, not general descriptors
Recyclability claims verified against actual recycling infrastructure in target markets
“Made in USA” or origin claims reviewed; qualified claims used where appropriate
Sustainability imagery (green colors, leaf icons, nature photography) reviewed for implied claim risk
Any third-party certification logos confirmed current and authorized for use on packaging
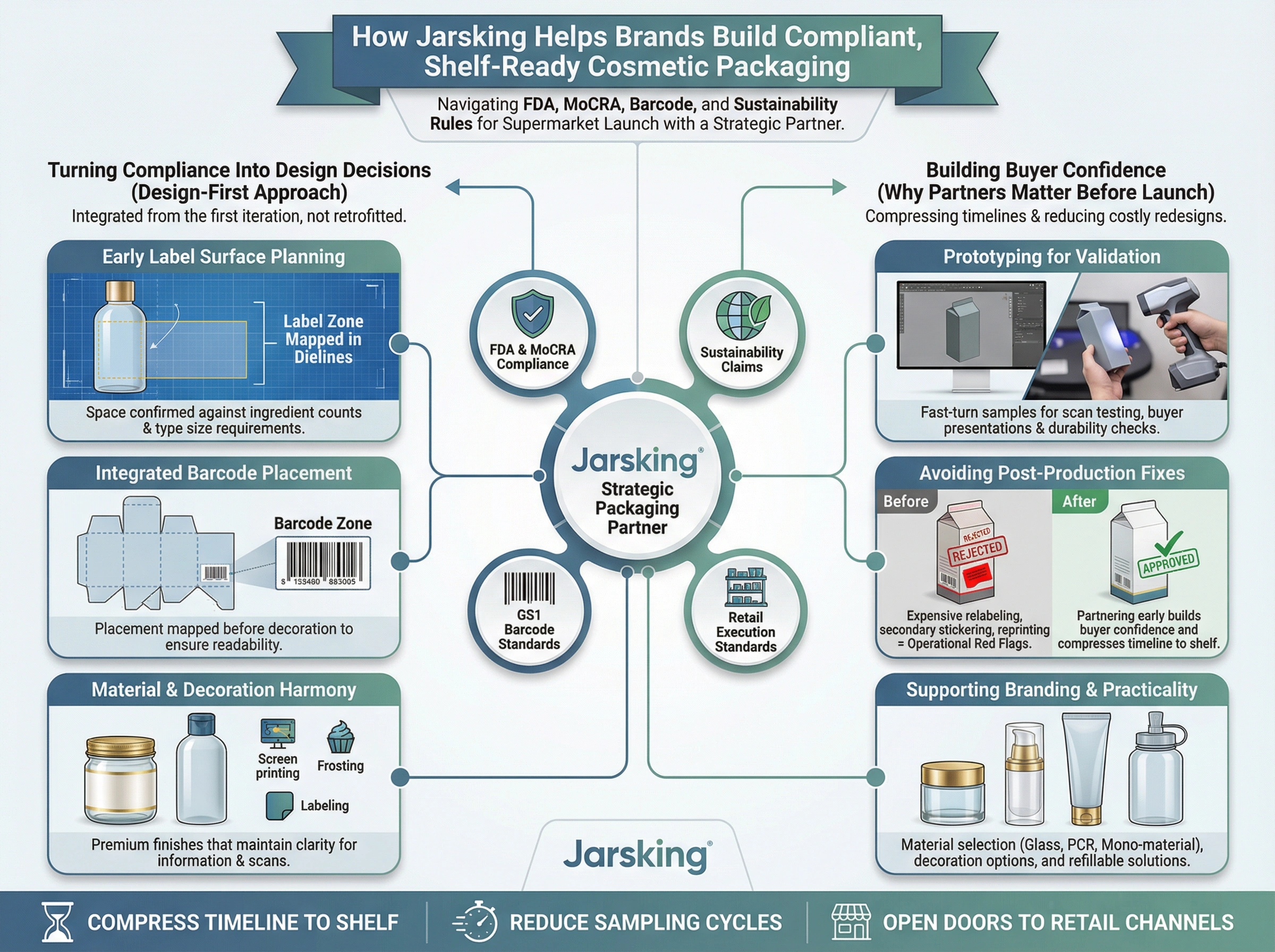
How Jarsking Helps Brands Build Compliant, Shelf-Ready Cosmetic Packaging
Navigating FDA requirements, MoCRA obligations, barcode standards, and sustainability claim rules simultaneously is a significant undertaking—especially for emerging brands and private-label founders approaching their first supermarket launch. That’s where an experienced cosmetic packaging manufacturer becomes a strategic partner, not just a supplier.
Turning Compliance Rules Into Packaging Design Decisions
Jarsking works across the full spectrum of cosmetic packaging formats—glass jars, plastic bottles, airless pumps, dropper bottles, tubes, roll-ons, and secondary cartons—with the structural expertise to integrate compliance requirements from the first design iteration rather than retrofitting them at the end.
This means label surface planning starts at the structural design stage, not after tooling is finalized. Barcode placement zones are mapped into dielines before decoration is applied. Ingredient panel space is confirmed against the actual ingredient count and type size requirements before samples are approved. The result is packaging that satisfies both FDA requirements and retail buyer expectations without requiring costly redesign cycles.
Supporting Both Premium Branding and Retail Practicality
The best cosmetic packaging for supermarkets doesn’t make you choose between compliance and brand equity. Jarsking’s capabilities span:
Material selection: Glass, PCR plastics, aluminum, and sustainable mono-material formats for brands making credible sustainability commitments
Decoration options: Screen printing, hot stamping, frosting, and labeling techniques that maintain barcode readability and ingredient panel clarity on premium finishes
Refillable systems: Structural solutions for refill programs that are growing in importance with both retail buyers and sustainability-focused consumers
Prototyping and sampling: Fast-turn physical samples for scan testing, retail buyer presentations, and distribution durability validation
Why Packaging Partners Matter Before Retail Launch
The cost of fixing a non-compliant package after production is far higher than the cost of getting compliance right before tooling is cut. Common post-production compliance fixes—relabeling runs, secondary stickering of missing information, reprinting cartons with corrected ingredient declarations—are expensive, time-consuming, and visible to buyers as operational red flags.
Working with a packaging partner who understands FDA cosmetic labeling requirements, MoCRA obligations, GS1 barcode standards, and retail execution standards from the start compresses your timeline to shelf, reduces sampling cycles, and builds the kind of buyer confidence that opens doors to additional retail channels over time.
Final Thoughts: Cosmetic Packaging Standards Are About More Than Compliance
The best cosmetic packaging for U.S. supermarkets does far more than satisfy a regulatory checklist. It communicates clearly to shoppers, scans reliably at checkout, holds up through the distribution chain, tells a credible brand story, and positions you for growth across multiple retail channels. That’s the bridge between compliance and competitive advantage.
U.S. cosmetic packaging requirements are shaped by the FDA’s labeling rules, MoCRA’s new safety and transparency obligations, FPLA’s net content and identity standards, and FTC’s claim guidelines—and all of them ultimately serve the same purpose: making sure consumers can trust what they’re buying. Brands that internalize that purpose, rather than treating compliance as a box-ticking exercise, build packaging that earns shopper trust and sustains long-term retail relationships.
Ready to build cosmetic packaging that’s compliant, shelf-ready, and brand-right for U.S. supermarkets? Contact Jarsking for a packaging consultation. Whether you need a retail-readiness review, a sustainable packaging solution, or a custom format built from scratch for your next supermarket launch, Jarsking’s team is ready to help you take it from concept to shelf-ready reality.
FAQs
U.S. cosmetic packaging for supermarkets must comply with FDA labeling rules (21 CFR 701 and 740), MoCRA obligations including adverse event contact information, Fair Packaging and Labeling Act disclosures, FTC claim standards, and GS1 barcode requirements. Packaging must also meet retail operational standards for durability, scan readability, and shelf presentation.
Required elements include a product identity statement and net quantity of contents on the principal display panel, plus the manufacturer or distributor’s name and address, a complete ingredient declaration in descending order of predominance, and applicable warning statements on the information panel. MoCRA additionally requires domestic contact information for adverse event reporting as of December 2024.
Yes. MoCRA requires cosmetic labels to include domestic contact information—an address, phone number, or electronic contact—for consumers and regulators to report adverse events. It also requires responsible persons to report serious adverse events to the FDA within 15 business days, and labels must exactly match the ingredient lists submitted during product registration.
Yes. Supermarkets require GS1-issued GTINs encoded in UPC-A (or UPC-E for small packages) format. Barcodes must be sourced directly from GS1 US to ensure globally unique numbers that pass retailer database verification. Third-party barcode resellers often provide recycled numbers that fail validation at major retailers.
GS1 US recommends placing retail barcodes in the lower right-hand section of the back of the package, with adequate quiet zones preserved on all sides. Avoid curved surfaces, seams, folds, and reflective finishes without conducting physical scan tests on actual production samples.
Yes. The FDA permits off-package ingredient labeling for packages under 12 square inches when the product is held in tightly compartmented trays or racks, is not enclosed in a folding carton, and cannot accommodate a full label. Strategies include secondary cartons, fold-out labels, hang tags, and display card disclosures.
Broad, unqualified claims like “eco-friendly,” “green,” “sustainable,” and “clean beauty” are risky without documented substantiation, as they may violate FTC Green Guides standards. Recyclability claims require verification that adequate recycling infrastructure actually exists in the target market. Origin claims using flags or nature imagery can imply environmental or “Made in USA” claims even without explicit text.
Start early: audit total label content requirements before finalizing structural design, confirm barcode specifications and placement with your manufacturer, review all claims against FTC Green Guides, and conduct physical scan tests on production samples before committing to full print runs. Partnering with a packaging manufacturer experienced in FDA cosmetic labeling requirements and retail-readiness standards—like Jarsking—reduces redesign cycles and accelerates your path to shelf.