Getting your cosmetic brand into Sephora, Ulta, or another prestige specialty retailer is one of the most powerful milestones a beauty founder can reach. These channels give brands immediate access to millions of engaged shoppers, strong editorial ecosystems, and in-store credibility that almost no other retail environment replicates. But earning that shelf placement—and keeping it—starts long before you pitch a buyer. It starts with your packaging.
Cosmetic packaging for Sephora and Ulta is evaluated on three simultaneous dimensions: regulatory compliance with FDA labeling standards, retailer-specific sustainability and ingredient programs, and aesthetic excellence that earns attention in one of the most visually competitive retail environments in the world. Brands that treat these as separate tasks—handling FDA compliance in one workstream and retailer program eligibility in another—almost always end up with costly redesigns, delayed vendor onboarding, or program rejections that push launches back by months.
This guide covers what every major U.S. specialty beauty retailer requires from cosmetic packaging. You’ll learn the exact thresholds behind Sephora’s Clean + Planet Aware seal, Ulta’s Conscious Beauty program, Credo Beauty’s industry-leading clean standard, and the specific operational requirements at Bluemercury, Space NK, and Sally Beauty. You’ll also see how working with an experienced packaging manufacturer like Jarsking lets you design packaging that satisfies every layer of those requirements while still looking unmistakably premium on shelf.
The Specialty Beauty Retail Landscape in the U.S.
Not all retail is equal for beauty brands. Supermarkets prioritize scan-readiness (see blog U.S. Cosmetic Packaging Standards for Supermarkets: A Practical Guide for Beauty Brands), net content accuracy, and distribution durability. Amazon demands FNSKU labels and poly-bagged leak protection. Specialty beauty retail is something else entirely—a channel where packaging aesthetics, sustainability credentials, and ingredient philosophy are evaluated with the same rigor as the product formula itself.
Why Specialty Beauty Retail Is a Different Kind of Packaging Challenge
Sephora, Ulta, and their peers are not passive distribution platforms. They are active brand curators. Each retailer uses its own proprietary programs—seals, badges, and certifications—to tier its assortment and reward brands that align with their values. A product that qualifies for Sephora’s Planet Aware seal receives premium shelf placement, digital badging, and editorial exposure that a non-qualifying product sitting three inches away will never see. Packaging that earns those designations doesn’t just satisfy a checklist—it becomes a commercial asset that drives sales velocity and deepens the retailer relationship.
For packaging teams, this means your design brief must carry two parallel sets of requirements from the very first conversation: the FDA’s legal labeling standards, and the retailers’ voluntary-but-effectively-mandatory program criteria that determine how prominently your brand gets featured.
The Six Specialty Beauty Retailers That Shape Packaging Standards
Here are the six retailers this guide covers, and the primary packaging dimension each one prioritizes:
| Retailer | Positioning | Primary Packaging Focus |
|---|---|---|
| Sephora | Prestige + mass prestige | Clean + Planet Aware seal programs, packaging material bans |
| Ulta Beauty | Mass prestige + drugstore | Conscious Beauty 5-pillar, PCR and recyclability thresholds |
| Credo Beauty | Strictest clean beauty | 2,700+ restricted ingredients, 50% PCR minimum, no single-use formats |
| Bluemercury | Luxury skincare boutique | Premium aesthetics, RangeMe onboarding, Macy’s distribution |
| Space NK | Curated luxury (U.S. + UK) | Premium materials, U.S. FDA adaptation for UK-origin brands |
| Sally Beauty | Professional & salon supply | UPC barcode compliance, “For Professional Use Only” labeling, bulk formats |
Sephora — Clean Programs, Packaging Material Bans, and Prestige Aesthetics
Sephora is the most influential specialty beauty retailer in the U.S. market, and its packaging program is correspondingly the most developed. Understanding Sephora’s three-seal framework is essential for any brand targeting prestige retail placement.
Sephora's Three Seals and What They Mean for Packaging
Sephora operates three distinct recognition programs, each with escalating requirements.
Clean at Sephora was launched in 2018 and has since grown to cover more than 3,000 products across 250+ brands. To qualify, products must be free from more than 50 restricted ingredient categories including parabens, SLS/SLES sulfates, phthalates, formaldehyde-releasing preservatives, mineral oil, and certain synthetic fragrances. In December 2025, Sephora extended the Clean program’s scope from formulation ingredients to packaging materials—restricting four categories from all Clean-designated packaging:
Halogenous compounds: PFAS, PFOS, PTFE, and related fluorinated chemicals
Bisphenols: BPA, PBDE, and related endocrine-disrupting compounds
Silver salts: Silver, silver chloride, and silver nitrate used as antimicrobial packaging additives
Mineral oil compounds: MOSH, MOAH, and related hydrocarbon contaminants
This means a brand’s packaging material composition—not just its formula—now determines Clean at Sephora eligibility.
Planet Aware recognizes brands with documented sustainable packaging commitments. Mandatory thresholds include:
75% of product packaging designed for circularity—recyclability, refillability, or compostability (50% for makeup brands)
Plastic packaging using at least 30% post-consumer recycled (PCR) content
Glass packaging made with at least 20% PCR content
Clean + Planet Aware is the highest-tier recognition, awarded only to brands that satisfy both programs simultaneously. It requires compliance across a minimum of 32 mandatory criteria spanning sustainable ingredient sourcing, sustainable packaging design, corporate commitments, and consumer transparency—and grants brands Sephora’s most visible editorial and in-store positioning.
What Sephora's Packaging Material Ban Means in Practice
The December 2025 material restrictions require brands to audit their packaging component supply chains in ways that go well beyond traditional formula review. Several common cosmetic packaging materials may now require replacement:
PTFE-coated pump mechanisms and closure liners must be verified PFAS-free
Polycarbonate components may contain bisphenols and should be replaced with PET, HDPE, or PP alternatives
Antimicrobial silver-additive packaging used in some “active” or “smart” packaging formats is now prohibited
Packaging printed with mineral-oil-based inks requires reformulation to UV-cured or water-based ink systems
Brands targeting Clean seal status must request material safety documentation—specifically Material Safety Data Sheets (MSDS) and component ingredient declarations—from every packaging supplier in their chain. This is an area where working with a manufacturer who proactively manages material compliance, like Jarsking, dramatically reduces the documentation burden at the buyer-onboarding stage.
Sephora's Operational and Aesthetic Packaging Expectations
Beyond program eligibility, Sephora sets clear expectations for how packaging performs operationally in its retail environment:
GS1-128 shipping labels are required for all cartons entering Sephora’s distribution centers
Counter-ready presentation matters: testers, display units, and secondary packaging must coordinate visually and structurally
Packaging consistency across SKU families is evaluated—a serum in a heavy glass dropper bottle and a moisturizer in a lightweight tube from the same brand need to look like they belong together on the shelf
Sephora’s Accelerate incubator program, which accepts applications annually and supports emerging indie brands, emphasizes packaging as a core component of commercial readiness. Brands that come to the program with compliant, sustainability-documented packaging in hand advance far faster than those who plan to address it after selection.
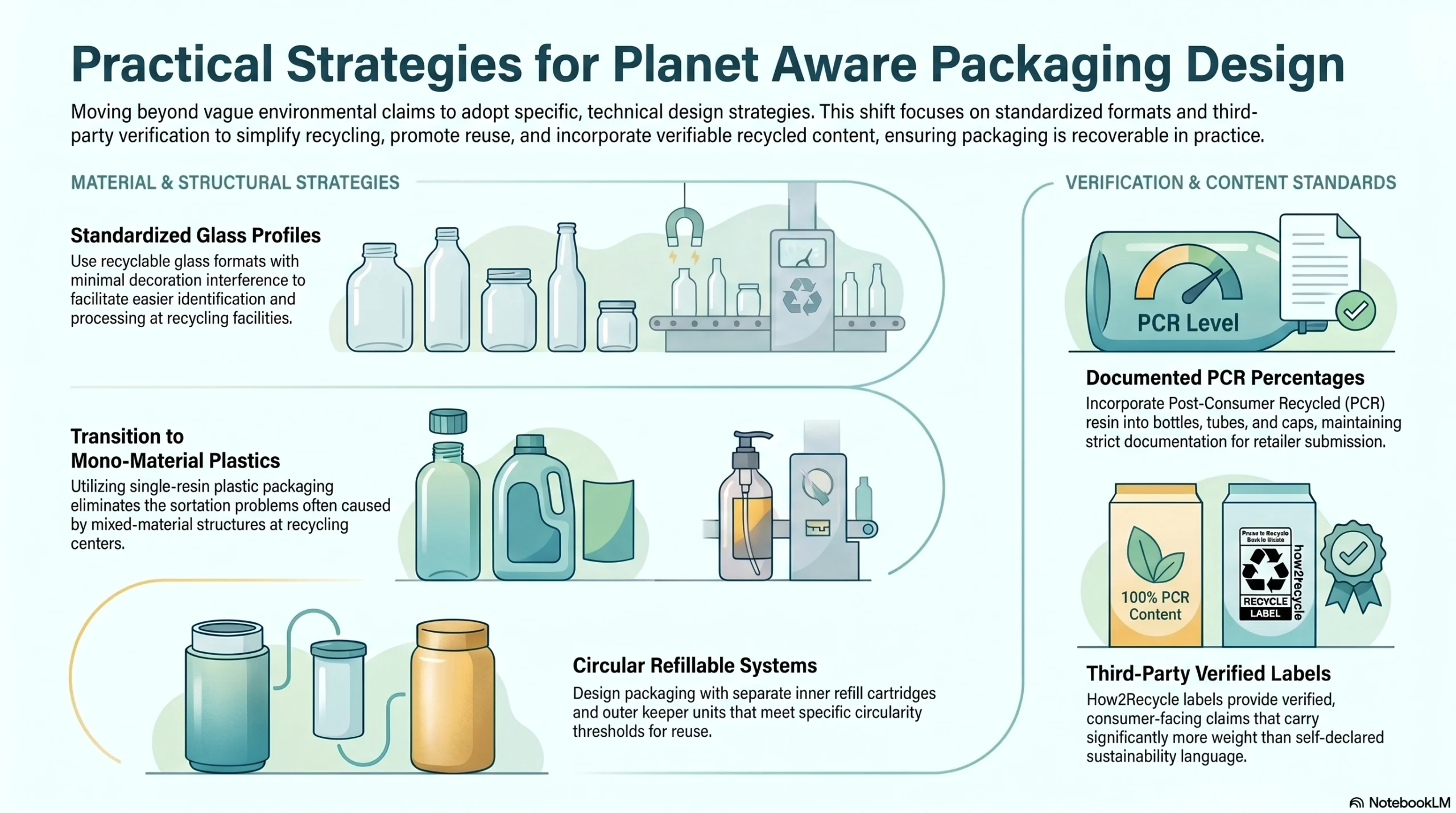
Practical Steps Toward Planet Aware Eligibility
Brands working toward Planet Aware qualification should prioritize these packaging design strategies:
Recyclable glass formats: Standardized glass profiles with minimal decoration interference facilitate easy recycling identification
Mono-material structures: Single-resin plastic packaging eliminates mixed-material sortation problems at recycling facilities
Refillable systems: Separate inner refill cartridges and outer keeper units that qualify under circularity thresholds
PCR plastics: Post-consumer recycled resin in bottles, tubes, and caps, with documented PCR percentages for retailer submission
Third-party certifications: How2Recycle labels provide verified, consumer-facing recyclability claims that carry significantly more weight than self-declared language
Ulta Beauty — Conscious Beauty's Five Pillars and Packaging Sustainability Thresholds
Ulta Beauty is the largest specialty beauty retailer in the U.S. by store count, with more than 1,400 locations. Its Conscious Beauty program creates a clear, shopper-visible sustainability tier that increasingly shapes which brands get featured in marketing, promotions, and editorial content.
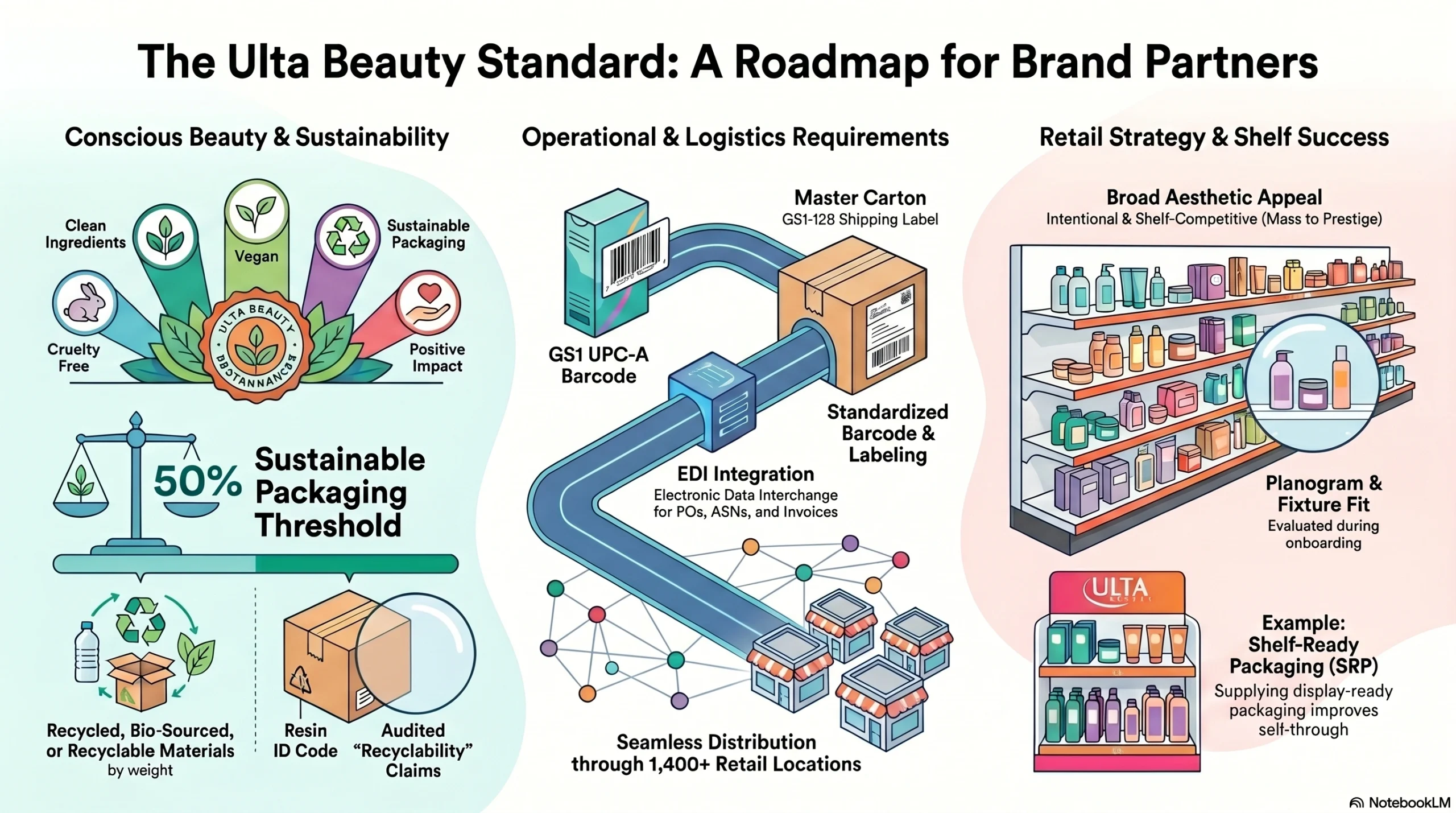
What Is the Conscious Beauty at Ulta Program?
Ulta's Sustainable Packaging Requirements
To qualify under the Sustainable Packaging pillar, at least 50% of a brand’s packaging by weight must use recycled, bio-sourced, or recyclable materials. Additional expectations include:
Resin ID codes must be present on all plastic packaging components
Refillable formats and reduced packaging weight are strongly encouraged
Packaging that misleads consumers about recyclability—including the use of “recyclable” claims for materials not accepted by mainstream U.S. recycling infrastructure—is flagged during compliance review
Brands participating in take-back programs (such as Pact Collective, which has a dedicated Sephora partnership) demonstrate additional circular commitment
Ulta's Vendor Logistics and Barcode Requirements
Ulta has specific operational requirements for all vendors supplying product through its distribution network:
UPC-A barcodes on every individual sellable unit, sourced from GS1 US
GS1-128 shipping labels on all master cartons entering Ulta distribution centers
EDI (Electronic Data Interchange) integration for purchase orders, advance shipping notices, and invoice processing
Shelf-ready packaging specifications may apply for floor displays and endcap placements
Ulta's Aesthetic Range vs. Sephora's
One meaningful difference between Ulta and Sephora is price-tier breadth. Ulta spans from drugstore-accessible price points to prestige skincare and fragrance—meaning packaging must read credibly to a far wider shopper demographic. A brand entering Ulta at a mid-market price point doesn’t need Sephora-level luxury packaging, but it does need to look intentional, clearly labeled, and shelf-competitive. Planogram fit and fixture compatibility are evaluated during buyer onboarding, and brands that supply their own display-ready packaging with consistent facings perform better during category resets.
Credo Beauty — The Industry's Strictest Clean and Packaging Standard
Credo Beauty operates 13 boutique stores across the U.S. and is widely recognized as setting the most rigorous clean beauty standard in the industry. For brands that qualify, Credo placement is a powerful signal of ingredient integrity and packaging responsibility that resonates strongly with the most ingredient-literate beauty consumers.
What Is the Credo Clean Standard?
The Credo Clean Standard restricts more than 2,700 ingredients—a figure that dramatically exceeds the FDA’s restricted list, Sephora’s Clean program, and the EU Cosmetics Regulation’s 1,300+ restricted substances. Key elements include:
Full INCI ingredient disclosure, including individual fragrance components above industry norms
Mandatory cruelty-free status—no animal testing at any stage of development or supply chain
No pesticide-contaminated ingredients that meet certain thresholds
Phthalate-free fragrance is an absolute requirement, not a program option
Credo's Sustainable Packaging Requirements
Credo’s packaging program is among the most detailed in the specialty beauty channel:
Minimum 50% PCR materials required for new brands entering the platform
All plastic packaging must carry resin ID codes (numbers 1–7 or their abbreviations) to facilitate consumer recycling
Prohibited formats: single-use masks, single-use wipes, single-serve sachets, and unnecessary decorative spatulas packaged with products
Smart design (right-sizing, reducing excess packaging, eliminating unnecessary components), sustainably sourced materials, and end-of-life optimization are the three foundational pillars of Credo’s packaging framework
No false or vague claims around recyclability or “green” materials—Credo expects specific, substantiated language
What Packaging Designers Must Know for Credo
Designing packaging for Credo requires material selection discipline at the component level. Virgin plastics should be reduced or eliminated wherever viable alternatives exist. Refillable and concentrated formats are strongly encouraged—both reduce packaging-to-product ratios and signal the brand’s sustainability intent. Decorative packaging choices—heavy embossing, mixed-material closures, foil-heavy cartons—that compromise recyclability or material sortation will not pass Credo’s packaging review without explicit justification. Documentation of PCR percentages, supplier certifications, and end-of-life pathways is expected before onboarding, not after.
Bluemercury — Luxury Skincare Boutique Packaging Standards
Who Bluemercury Is and Who It's Right For
How to Submit Products to Bluemercury
All vendor submissions to Bluemercury are handled through the RangeMe platform, which Macy’s uses across its buying organization for new product discovery. This means packaging must photograph cleanly and represent the brand accurately in a digital submission format before it is ever seen by a buyer in person.
Bluemercury does not publish a detailed public packaging sustainability program the way Sephora or Credo does. However, the quality of materials, premium finishes, and overall brand coherence are evaluated by buyers who are deeply familiar with the luxury skincare segment. Packaging that looks misaligned with Bluemercury’s positioning—regardless of formula quality—is unlikely to advance past the initial submission stage.
Packaging Aesthetic Requirements for Luxury Boutique Retail
For brands targeting Bluemercury, packaging design priorities include:
Premium structural materials: Weighted glass, metal closures, and matte or frosted finishes read as luxury in this context
FDA labeling in refined layouts: Required disclosures must be present and legible—but in a format and type treatment that maintains the premium brand presentation
Counter display coordination: Testers, display cards, and travel-size counterparts must be designed as a coordinated family
Ingredient transparency: While Bluemercury doesn’t operate a formal restricted ingredient program, its shoppers expect clear ingredient communication and clean-leaning formulation narratives
Space NK — Curated Luxury Retail for Niche and International Brands
Space NK is a UK-founded prestige beauty retailer with a growing U.S. presence, including standalone stores in major coastal markets and shop-in-shop locations within Bloomingdale’s. It is particularly relevant for niche European and UK brands seeking a curated U.S. retail entry point, and for American indie brands looking to reach a fashion-forward, luxury-oriented consumer.
Space NK's Positioning and U.S. Presence
Space NK is known for stocking innovative, trend-forward brands that aren’t typically found in mass prestige retail. Its edit is deliberately tight—which means packaging must not just comply but impress. Premium aesthetics, ingredient integrity, and a distinctive brand point of view are all evaluated as part of the buyer conversation. The retailer’s shopper expects to discover something unfamiliar, which gives niche packaging concepts room to succeed here that might struggle at a more mainstream channel.
Key Packaging Considerations for UK Brands Entering the U.S. Through Space NK
This is one of the most practically important sub-sections for brands in Space NK’s typical pipeline. UK and EU-origin brands often assume their existing packaging translates directly to U.S. retail—it does not. Key adaptations required include:
Net quantity in U.S. customary units: Fluid ounces and ounces avoirdupois must appear on the principal display panel; metric equivalents may follow as secondary
FDA ingredient declaration format: EU Cosmetic Regulation allows ingredients listed by INCI name in descending order—which aligns with FDA requirements—but EU packages often omit the level of specificity FDA requires for certain ingredient sub-categories
MoCRA adverse event contact information: A domestic U.S. address, phone number, or electronic contact must appear on the label—this obligation does not exist under EU regulation and is almost always missing on unmodified EU packaging
Warning statements: FDA-required warnings differ materially from EU warnings; aerosol, flammable, and sunscreen OTC warnings must all be reviewed specifically for FDA compliance
“Made in” origin statements: UK or EU origin claims must comply with FTC standards when the product is marketed in the U.S.
Brands that plan their U.S. packaging adaptation early—ideally in parallel with the Space NK buyer conversation rather than after it—avoid the rushed, expensive last-minute label reprints that delay launches.
Sally Beauty — Professional Channel Packaging and Compliance
Sally Beauty is the largest professional beauty supply retailer in the United States, with over 4,600 stores serving both licensed professionals and everyday consumers. Its packaging context is fundamentally different from every other retailer in this guide—functional communication, professional-use labeling, and operational logistics compliance take clear priority over aesthetic luxury.
Sally Beauty's Market Position and Packaging Context
Sally Beauty’s assortment spans hair color, chemical services, professional tools, skincare, and nail. Products that cross into OTC drug territory—professional-strength hair treatments, chemical relaxers, certain nail products—face FDA drug labeling requirements including Drug Facts panels, in addition to standard cosmetic labeling rules. This dual-channel nature (consumer retail + professional supply) means packaging must be designed to communicate clearly across two audiences with different levels of technical expertise.
Sally Beauty's Vendor Packaging Requirements
Sally Beauty publishes detailed Vendor Performance Requirements that all new suppliers must meet:
All individual sellable units must carry a scannable UPC-A barcode printed on or affixed to the item
Packing slips included in each master carton must specify UPC code, vendor item number, and product description for every unit inside
Shelf life at time of receipt must exceed 12 months for all cosmetic products—a requirement that affects how brands manage production lead times and inventory rotation
“For Professional Use Only” declarations are required on all products intended for licensed professional application
Bulk or professional-size formats require accurate net quantity labeling that reflects the professional-use context
Packaging Design for the Professional Beauty Channel
In the professional channel, packaging hierarchy shifts significantly compared to prestige retail. Safety information, usage instructions, application ratios, and professional credential references move to primary label positions. Color-coding systems are widely used to differentiate product lines within professional categories—permanent vs. demi-permanent color, developer volumes, treatment intensity levels. Structural formats tend toward functional rather than luxury: flip-top caps over dropper pumps, professional-size tubes over single-use formats, clearly printed volume markings on liquid containers.
Shared Packaging Requirements Across All Specialty Beauty Retailers
Despite the significant differences between Sephora’s Planet Aware program and Sally Beauty’s vendor barcode requirements, every specialty beauty retailer in the U.S. operates within the same federal regulatory framework. These requirements are non-negotiable regardless of which channel you target.
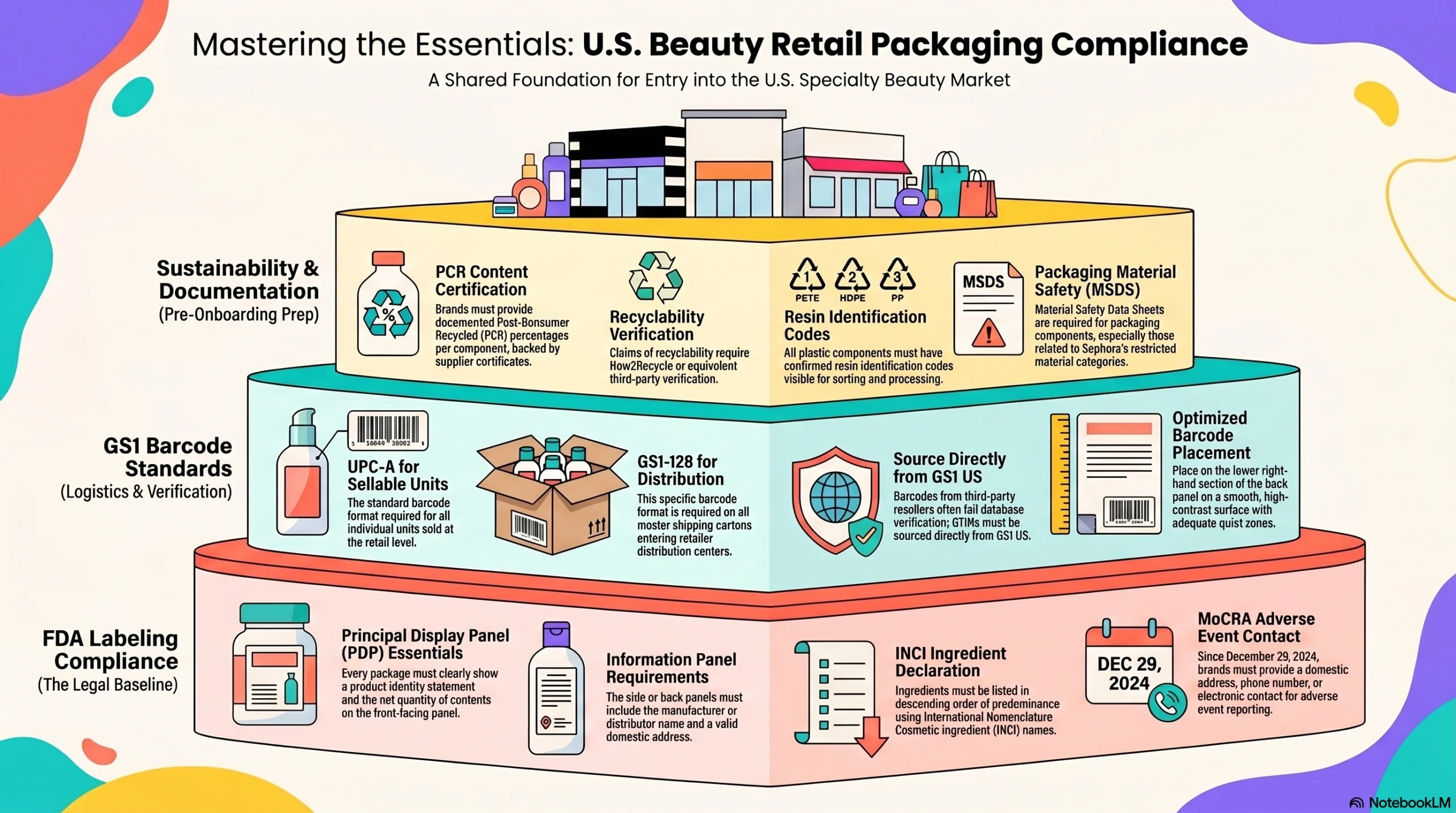
FDA Labeling Compliance Is Non-Negotiable Across All Channels
Every cosmetic sold through any U.S. retailer must comply with FDA labeling requirements under 21 CFR Parts 701 and 740. That means every package—from a Credo boutique launch to a Sally Beauty professional SKU—must carry:
A product identity statement and net quantity of contents on the principal display panel
Manufacturer or distributor name and domestic address on the information panel
Complete ingredient declaration in descending order of predominance using INCI names
Required warning statements where applicable
MoCRA adverse event contact information: a domestic address, phone number, or electronic contact, required since December 29, 2024
Retailer sustainability programs don’t supersede these requirements—they layer on top of them.
GS1 Barcodes Are Required Across Every Specialty Retailer
Every retailer in this guide requires GS1-issued barcodes for retail units and distribution cartons. UPC-A is the standard for individual sellable units; GS1-128 is required on master shipping cartons entering retailer distribution centers. GTINs must be sourced directly from GS1 US—barcodes purchased from unauthorized third-party resellers frequently fail retailer database verification during vendor onboarding. Barcode placement should follow GS1 US guidance: lower right-hand section of the back panel, with adequate quiet zones, on a smooth, high-contrast surface.
Sustainability Documentation Is Expected Before Onboarding
Brands targeting Sephora, Ulta, or Credo should prepare sustainability documentation packages before initiating buyer conversations. This includes:
Documented PCR content percentages per packaging component, with supplier certificates
How2Recycle or equivalent recyclability verification where recyclable claims are made
Resin identification codes confirmed on all plastic components
Material Safety Data Sheets covering packaging components for Sephora’s restricted material categories
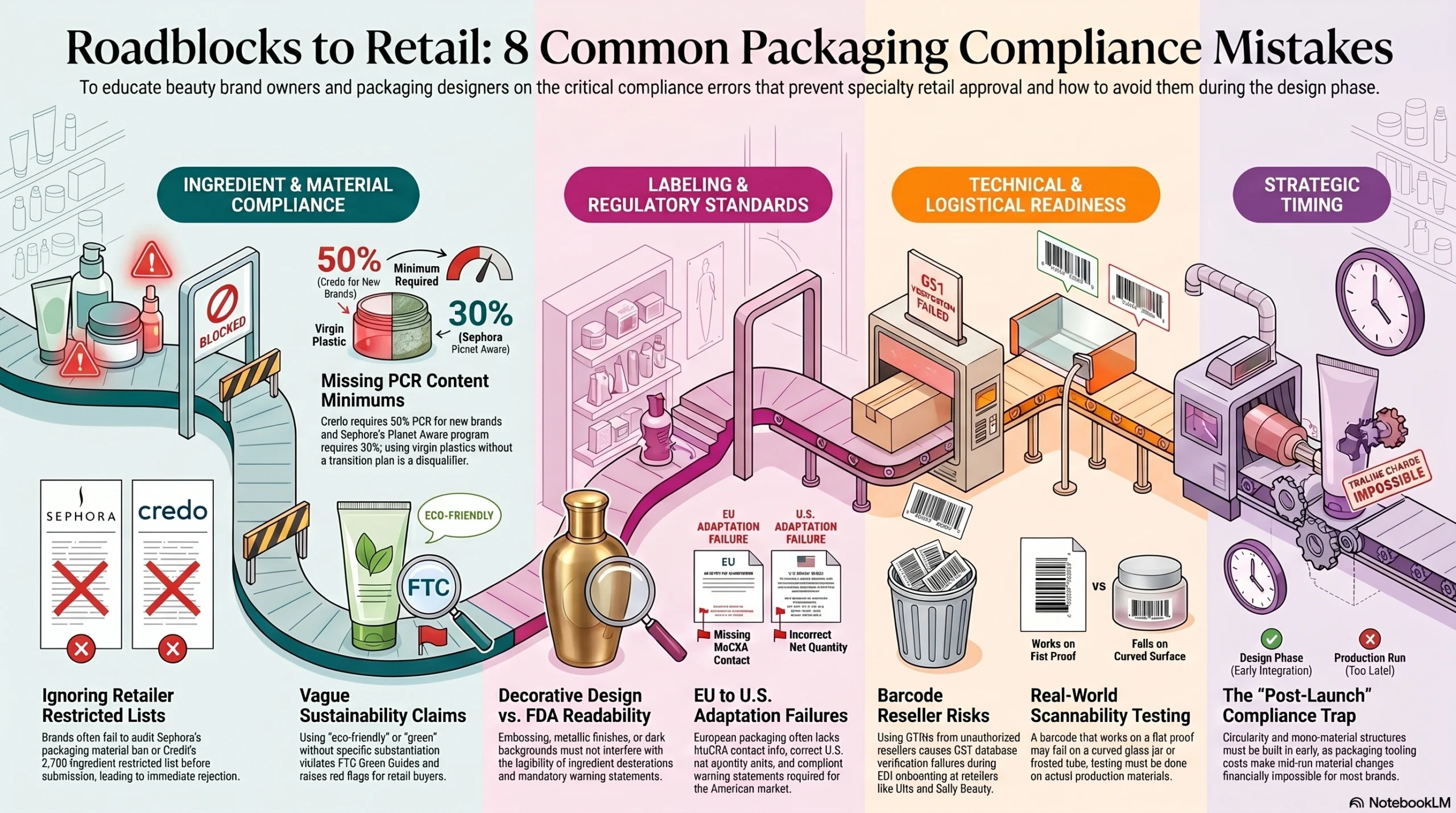
Common Packaging Mistakes That Prevent Specialty Retailer Approval
These are the most frequent packaging errors that delay or block specialty beauty retail entry:
Submitting packaging before auditing the retailer’s restricted ingredient and materials list — Sephora’s packaging material ban and Credo’s 2,700-ingredient restricted list are both publicly available; there is no excuse for discovering conflicts at the onboarding stage
Using virgin plastics without a documented PCR transition plan — Credo requires 50% PCR for new brands; Sephora’s Planet Aware program requires 30% PCR for plastics; brands with no PCR strategy are ineligible from the start
Making broad sustainability claims without specific language — “eco-friendly,” “green,” and “sustainable” packaging claims without substantiation violate FTC Green Guides standards and raise red flags with sustainability-focused retail buyers
Designing decorative packaging that compromises FDA label readability — embossed surfaces, dark backgrounds, and metallic finishes must still permit conspicuous, legible ingredient declarations and warning statements
Using GTINs from unauthorized barcode resellers — these numbers fail GS1 database verification at Ulta, Sally Beauty, and every other retailer using EDI vendor onboarding
Launching U.S. packaging adapted from EU SKUs without FDA adjustments — missing MoCRA contact information, incorrect net quantity units, and non-compliant warning statements are the three most common failures
Failing to test barcode scannability on actual production material — a barcode that scans perfectly on a flat proof may fail 30% of scan attempts on a curved glass jar or frosted tube
Treating sustainability program compliance as a post-launch task — packaging tooling costs make mid-run material changes prohibitively expensive; PCR content, mono-material structures, and circularity design must be built in at the structural design stage
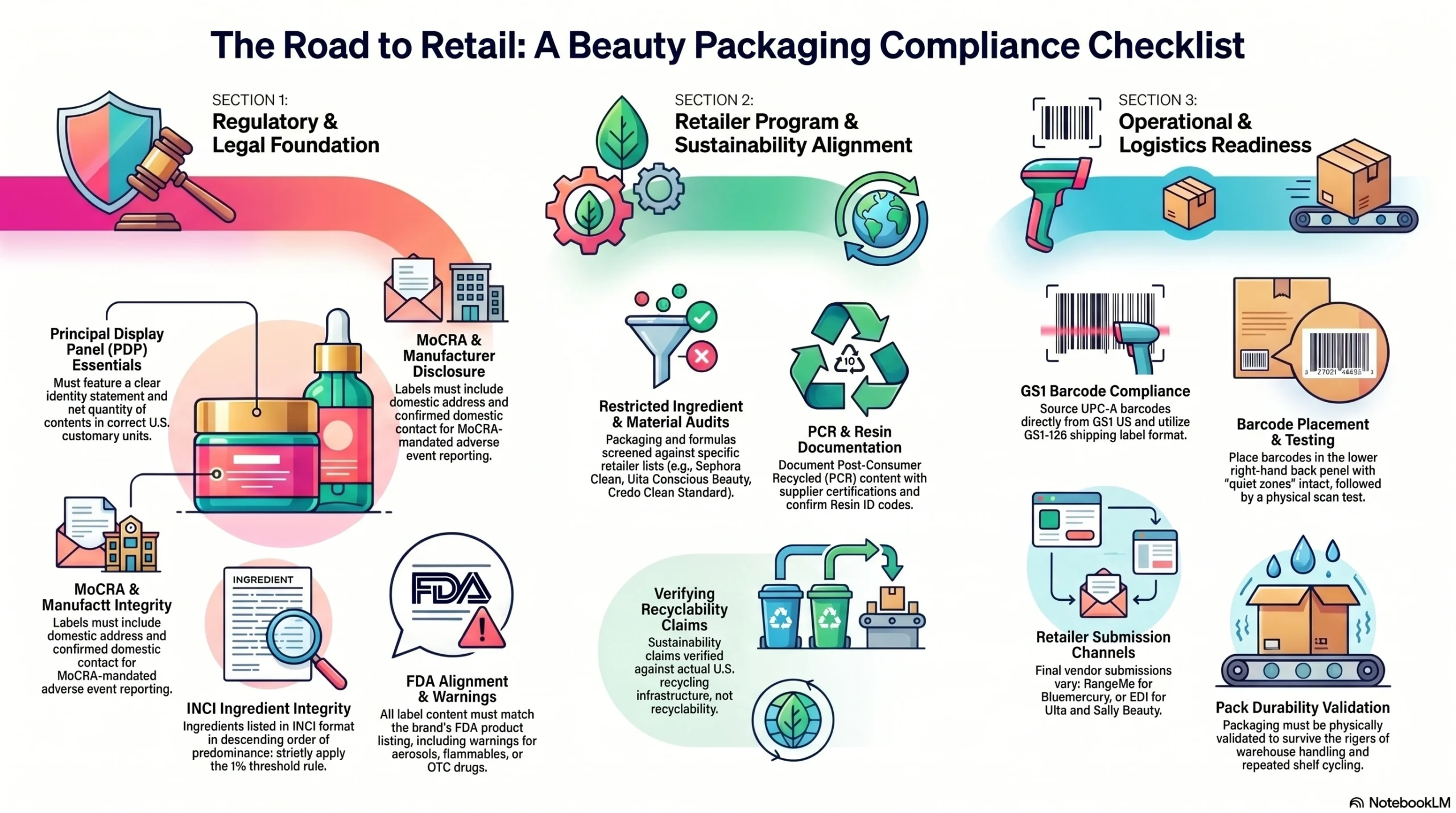
Packaging Checklist for Specialty Beauty Retail Entry
Use this checklist during your packaging development process, before committing to production tooling.
Regulatory Compliance Checklist
Product identity statement and net quantity on principal display panel, in correct U.S. customary units
Manufacturer or distributor name and domestic address on information panel
MoCRA adverse event domestic contact information confirmed on label
Ingredient declaration in INCI format, descending order of predominance, with 1% threshold rule applied
Required warning statements reviewed and included (aerosol, flammable, OTC drug where applicable)
Label content matches FDA product listing submission
Retailer Program Eligibility Checklist
Restricted ingredient list reviewed against target retailer’s program (Sephora Clean, Ulta Conscious Beauty, or Credo Clean Standard)
Packaging material composition audited against Sephora’s four prohibited material categories (if targeting Clean seal)
PCR content percentage documented with supplier certification
Recyclability claims verified against real-world U.S. recycling infrastructure availability
Resin ID codes confirmed on all plastic components
Refillable format assessed for program eligibility where applicable
Operational and Logistics Checklist
UPC-A barcode sourced directly from GS1 US
GS1-128 shipping label format confirmed for retailer distribution centers
Barcode placement in lower right-hand section of back panel, with quiet zones preserved
Physical scan test conducted on production sample—not just digital proof
Pack durability validated for retailer warehouse handling and shelf cycling
Vendor submission completed via required channel: RangeMe for Bluemercury, EDI for Ulta and Sally Beauty
How Jarsking Helps Brands Win Specialty Beauty Retail Placement
Navigating FDA compliance, retailer sustainability programs, material documentation requirements, and luxury aesthetic standards simultaneously is a significant undertaking. It’s especially demanding for emerging brands approaching their first Sephora pitch or Credo submission. That’s where an experienced cosmetic packaging manufacturer becomes a strategic partner—not just a supplier.
Packaging That Satisfies Both the Regulator and the Retailer
Jarsking works across the full spectrum of cosmetic packaging formats—PCR glass jars, recycled plastic bottles, airless pumps, refillable systems, luxury cartons, and secondary display packaging—with the structural and material expertise to integrate compliance requirements from the very first design iteration.
This means label surface planning starts at the structural design stage, not after tooling is finalized. PCR content percentages are documented and provable before retailer submission. Material safety documentation for Sephora’s restricted material categories is prepared in parallel with structural sampling. Barcode placement zones are mapped into dielines before any decoration is applied. The result is packaging that enters buyer conversations already qualified—rather than requiring mid-process revisions that compress timelines and inflate cost.
Sustainable Packaging Formats That Qualify for Retailer Programs
Jarsking’s sustainable packaging capabilities align directly with the requirements of Sephora’s Planet Aware, Ulta’s Conscious Beauty, and Credo’s packaging framework:
PCR glass and recycled plastic options with documented post-consumer recycled content percentages
Mono-material structures designed for clean recyclability across standard U.S. sortation systems
Refillable systems with separate inner refill cartridges and outer keeper units engineered for multiple fill cycles
Right-sized packaging that reduces material weight without compromising structural integrity or shelf presence
Packaging formats compatible with Pact Collective and similar take-back programs
Premium Aesthetics That Meet Luxury Retail Standards
Sustainability and premium aesthetics are not in conflict—but they require deliberate engineering to coexist. Jarsking’s decoration capabilities—hot stamping, frosting, matte lacquering, screen printing, and embossing—are available on packaging structures that are simultaneously PCR-compliant, barcode-readable, and FDA-labeled. Counter display systems, coordinated tester packaging, and travel-size counterparts are engineered together rather than as afterthoughts, giving brands the visual coherence that Sephora, Bluemercury, and Space NK buyers expect.
Prototyping and fast-turn sampling allow brands to put physical samples in buyers’ hands—and conduct scan tests on actual production materials—before committing to full production runs.
Final Thoughts: Specialty Retail Packaging Is a Strategic Investment, Not a Cost Center
The best cosmetic packaging for Sephora, Ulta, Credo, and their peers does far more than meet a compliance checklist. It communicates a brand’s ingredient philosophy before a shopper reads a single word. It signals sustainability credibility before a buyer asks for documentation. It earns program eligibility, editorial placement, and shopper trust simultaneously—and that’s the real commercial return on packaging investment in specialty beauty retail.
Brands that plan for regulatory compliance, retailer program requirements, and luxury aesthetics together—from the first structural brief—compress their timeline to shelf, reduce expensive redesign cycles, and walk into buyer meetings with packaging that already makes the right argument.
Ready to build specialty beauty retail–ready packaging? Contact Jarsking for a packaging consultation. Whether you need PCR material documentation for a Sephora Planet Aware submission, a refillable format designed for Credo eligibility, or premium glass packaging engineered for Bluemercury’s luxury counter, Jarsking’s team is ready to take your packaging from concept to shelf-ready reality.
FAQs
Sephora requires FDA-compliant labeling on all products, plus program-specific requirements for brands seeking Clean at Sephora, Planet Aware, or Clean + Planet Aware seal status. As of December 2025, Clean at Sephora extends to packaging materials—prohibiting PFAS, bisphenols, silver salts, and mineral oil compounds in packaging components. Planet Aware requires 30% PCR for plastics, 20% PCR for glass, and 75% of packaging designed for circularity.
Ulta’s Conscious Beauty Sustainable Packaging pillar requires that at least 50% of a brand’s packaging by weight use recycled, bio-sourced, or recyclable materials. Resin ID codes must be present on all plastic components, and recyclability claims must reflect genuine U.S. recycling infrastructure availability.
Credo requires a minimum of 50% PCR materials for new brands, resin ID codes on all plastics, and prohibits single-use formats including sachets, single-use wipes, and single-use masks. Full INCI disclosure, cruelty-free status, and no vague or unsubstantiated sustainability claims are also mandatory.
Yes, without exception. FDA labeling requirements—including product identity, net quantity, ingredient declaration, manufacturer details, and MoCRA adverse event contact information—apply to all cosmetics sold in the U.S., regardless of which retailer carries them. Retailer sustainability programs layer additional requirements on top of, not instead of, federal law.
Packaging that qualifies includes glass with 20%+ PCR content, plastics with 30%+ PCR content, mono-material structures designed for recyclability, and refillable formats. Third-party verification such as How2Recycle labels strengthens program submissions.
Credo restricts over 2,700 ingredients versus Sephora’s 50+ Clean ingredient list, and its 50% PCR minimum for new brands exceeds Sephora’s 30% Planet Aware threshold for plastics. Credo also prohibits specific single-use packaging formats entirely—something Sephora’s program does not explicitly mandate.
No. EU-origin packaging typically lacks MoCRA adverse event contact information, uses metric-only net quantity labeling, and may carry EU-specific warning statements that don’t satisfy FDA requirements. FDA-compliant U.S. packaging adaptations must be completed before any U.S. retail launch, including Space NK placement.
All specialty beauty retailers require UPC-A barcodes on individual sellable units, sourced directly from GS1 US. GS1-128 labels are required on master shipping cartons for distribution center receiving at Sephora, Ulta, and Sally Beauty. Barcodes from unauthorized third-party resellers fail retailer database verification and will block vendor onboarding.